Diagnostic Microbiology — MCQs

On this page
What is incorrect about the media shown below?

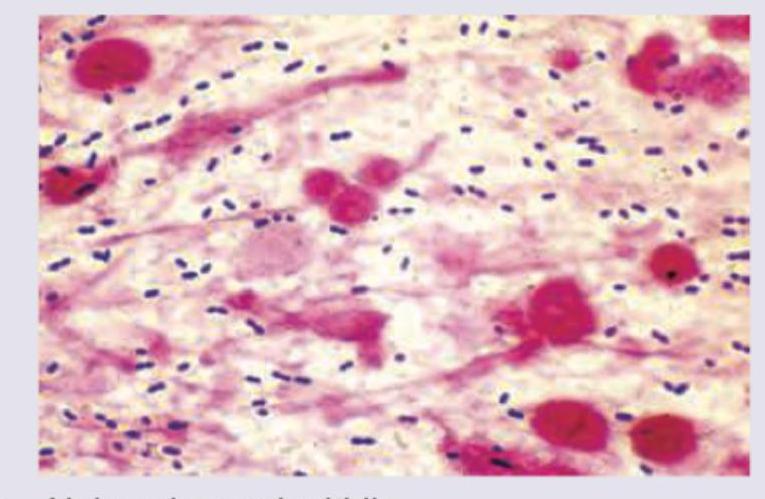
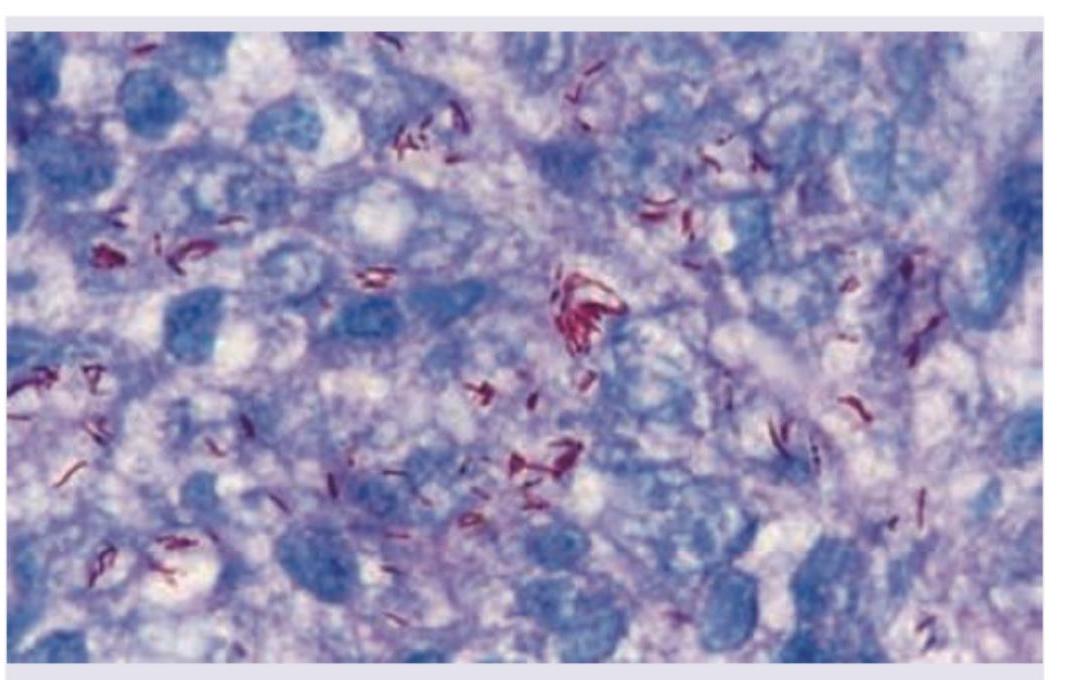
The gram stain given below shows which organism most likely? (AIIMS May 2016)
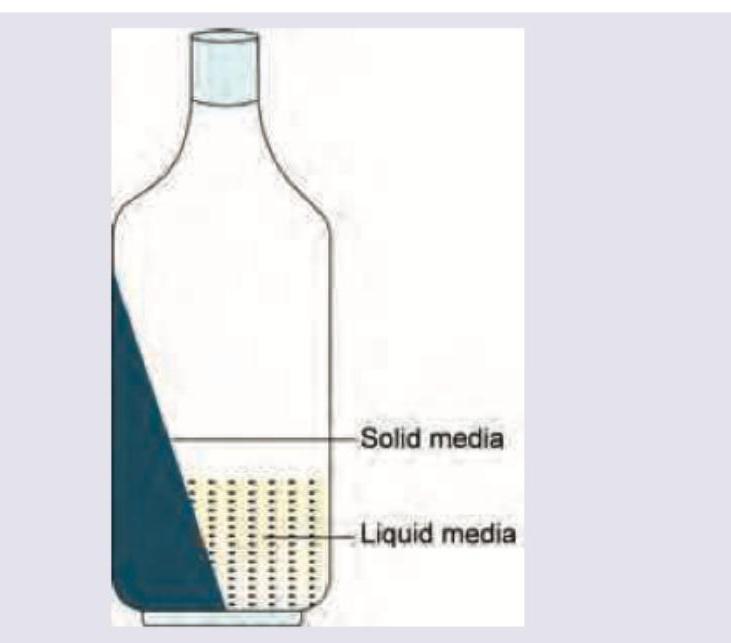
The following two phase culture system is used for diagnosis of: (Recent NEET Pattern 2016-17)
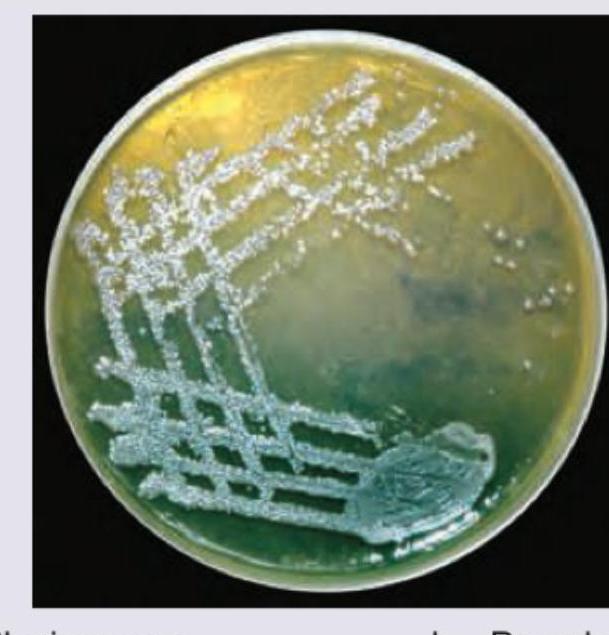
Which of the bacteria is shown in this culture plate of nutrient agar? (Recent NEET Pattern 2016-17)
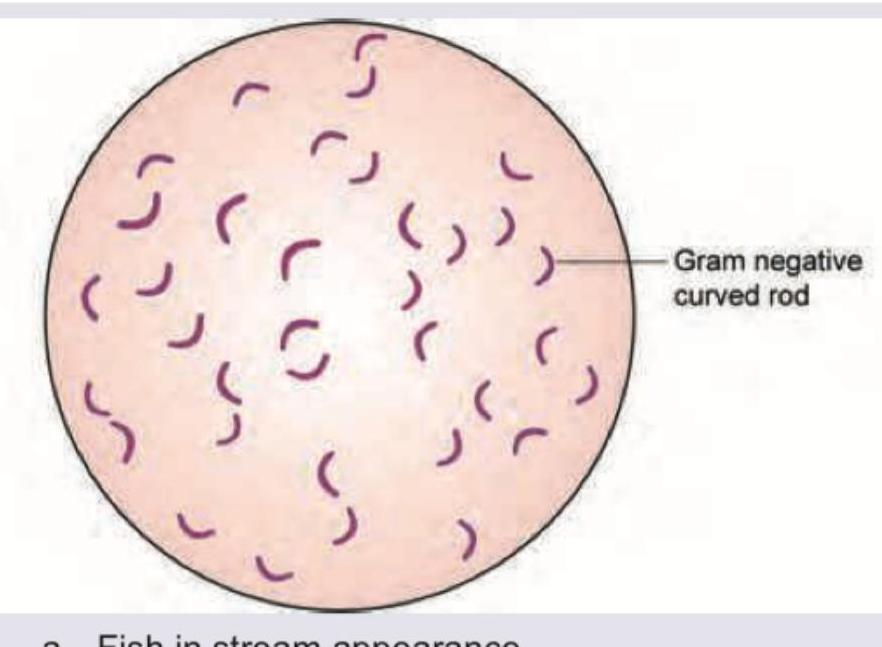
All are correct about the organism shown except:
All are true about the organism shown except:
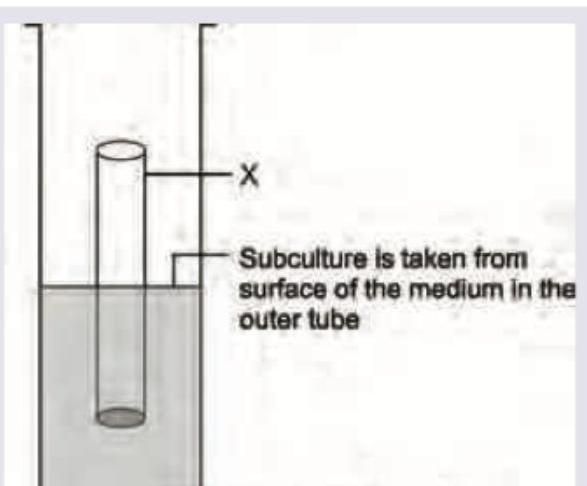
Identify the tube marked as X in culture of Salmonella typhi.
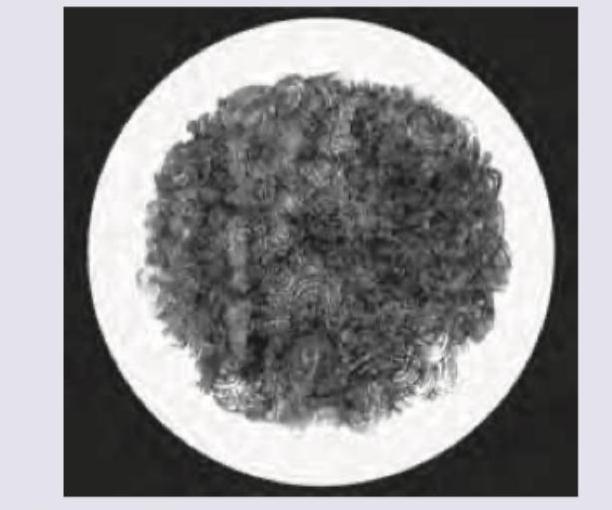
What is the drug of choice for organism producing the following colonies?
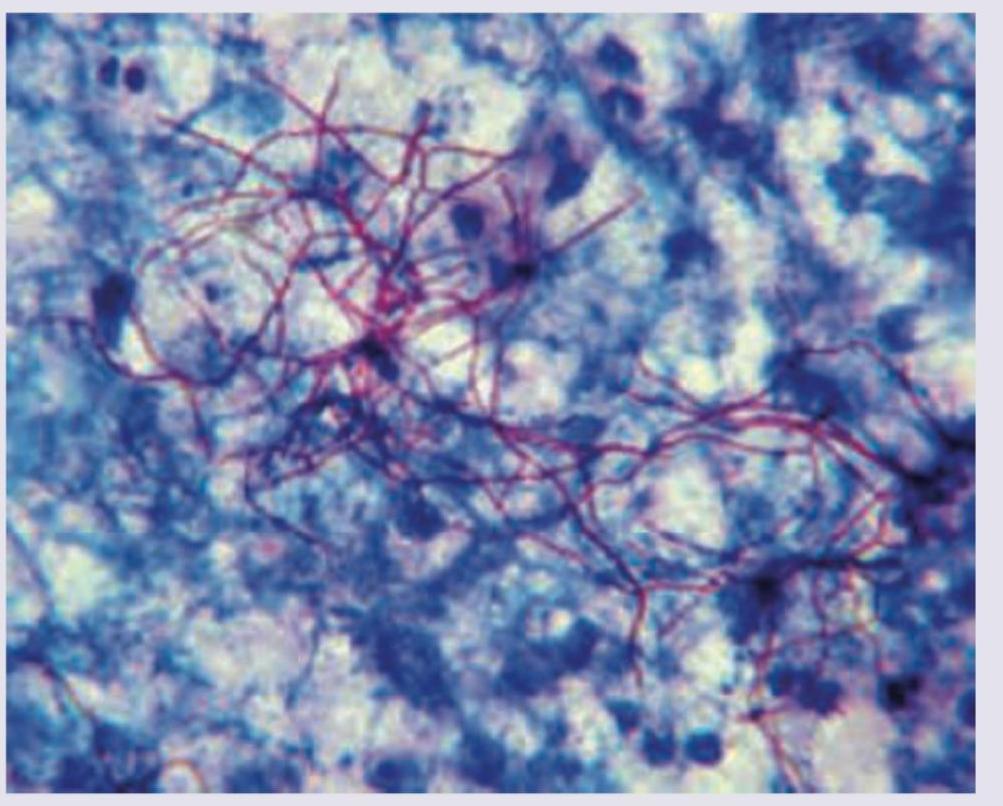
Identify the organism shown below. (AIIMS May 2017)
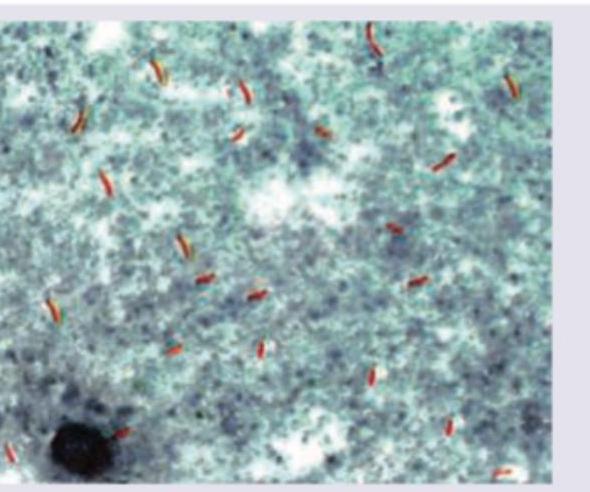
Identify the organism seen in the slide shown below.
Practice by Chapter
Specimen Collection and Transport
Practice Questions
Microscopy in Microbiology
Practice Questions
Culture Methods and Media
Practice Questions
Bacterial Identification Techniques
Practice Questions
Antimicrobial Susceptibility Testing
Practice Questions
Serological Diagnosis
Practice Questions
Molecular Diagnostic Methods
Practice Questions
Rapid Diagnostic Tests
Practice Questions
Point-of-Care Testing
Practice Questions
Automation in Microbiology Laboratory
Practice Questions
Quality Control in Diagnostic Microbiology
Practice Questions
Interpretation of Microbiological Reports
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
