Bacteriology — MCQs

On this page
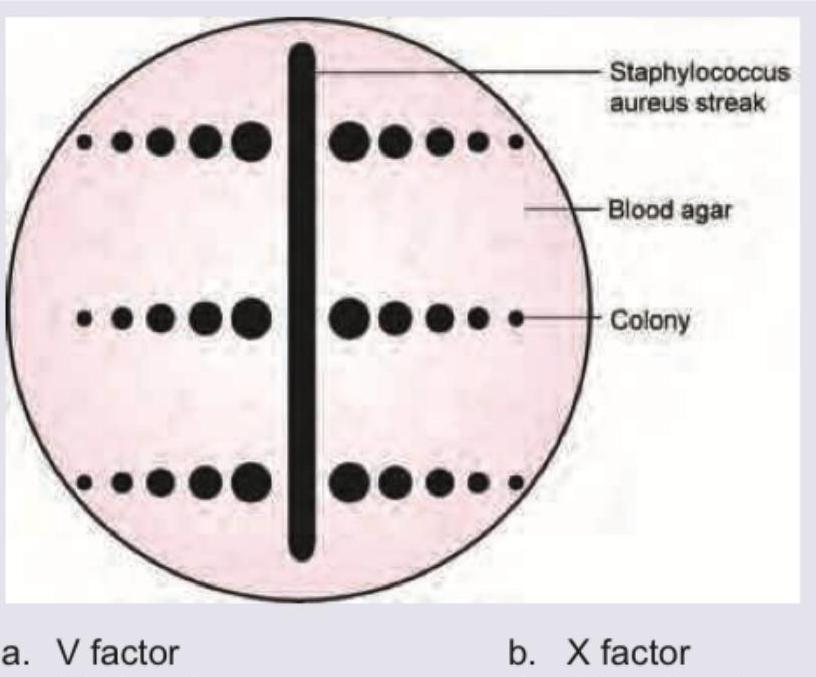
The following plate shows *H. influenzae* colonies. Which of the following is responsible for the phenomenon shown? (Recent NEET Pattern 2016-17)
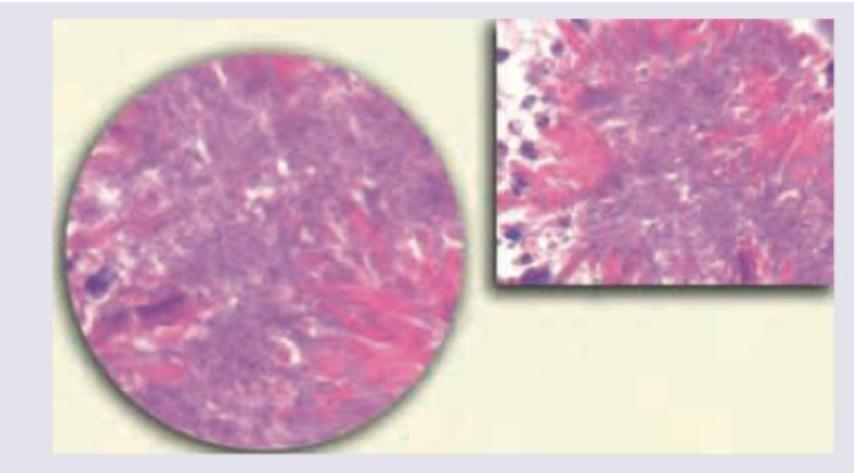
A 50-year-old village agricultural worker got a dental treatment from a quack for painful dental caries. He later developed a cheek swelling with pus discharge from sinus containing yellow specks. The specks were crushed between two slides and stained with gram stain and ZN stain. All are true about the incriminating organism except:
The image given below shows:

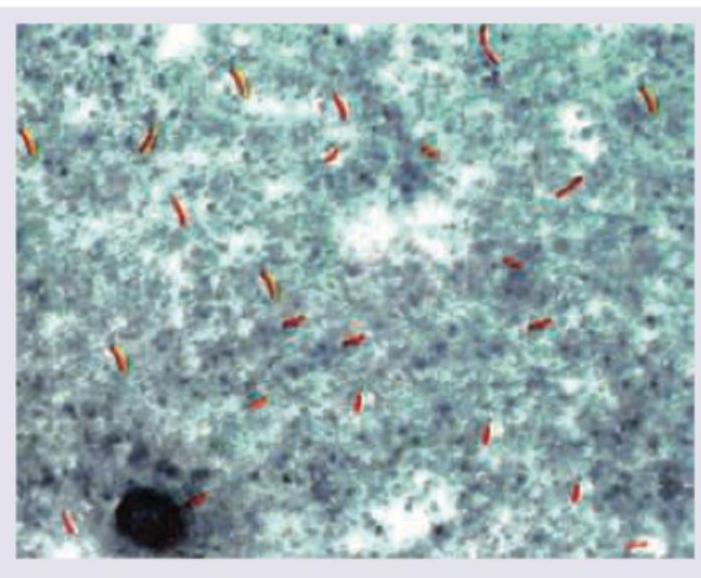
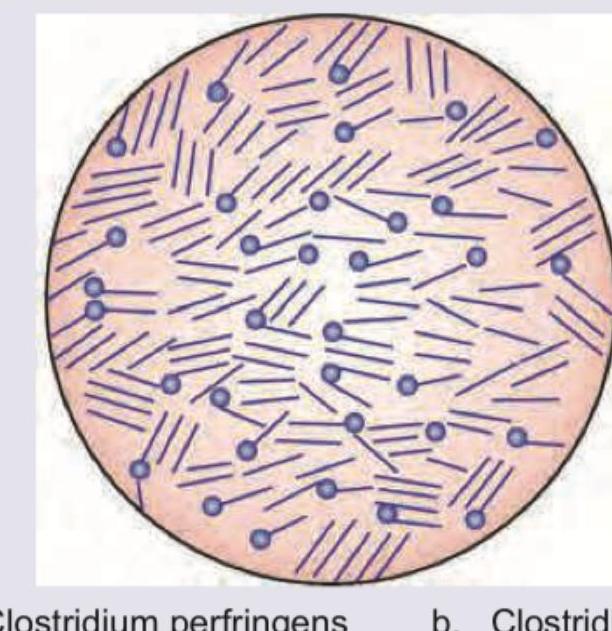
All are correct about the organism shown below except:
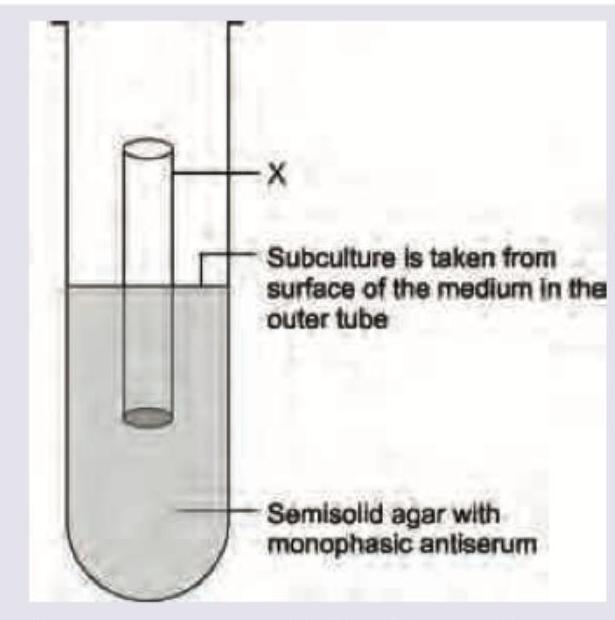
The component marked X in Salmonella typhi culture is used for determination of:
All are true about the organism shown except:
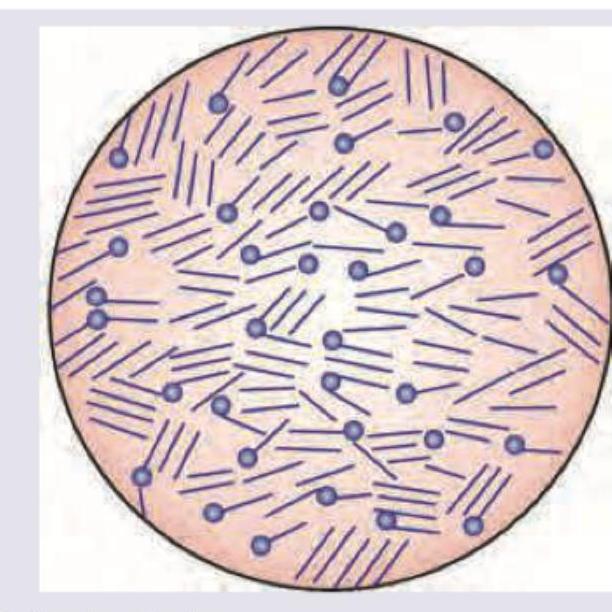
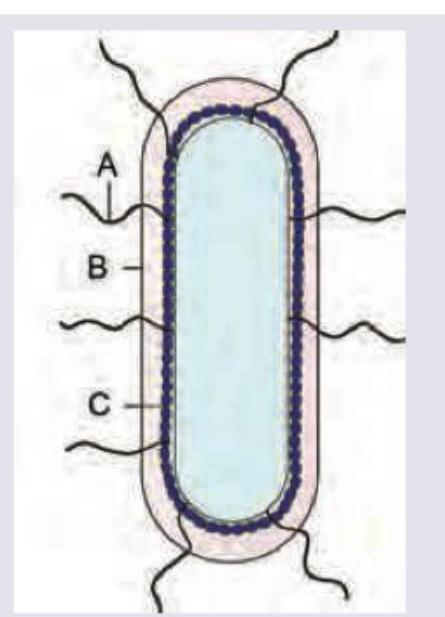
What is correct about the schematic image of Salmonella typhi?
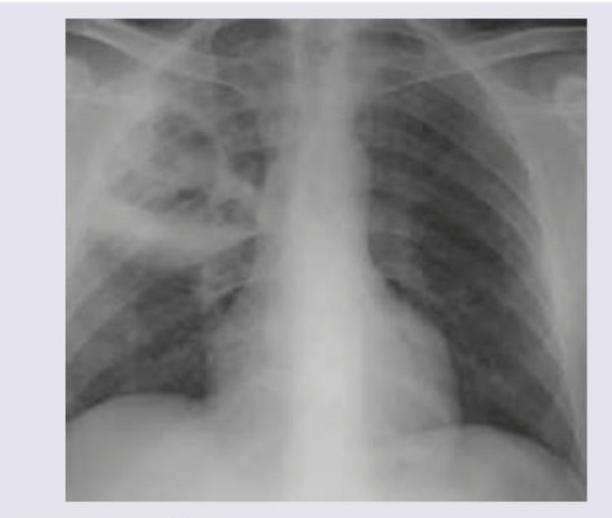
A 45-year-old alcoholic presents with severe respiratory distress and red current jelly sputum. On examination bronchial breathing was heard in right infraclavicular and inframammary areas. The X-ray chest is shown below. All are correct about the organism incriminated except:
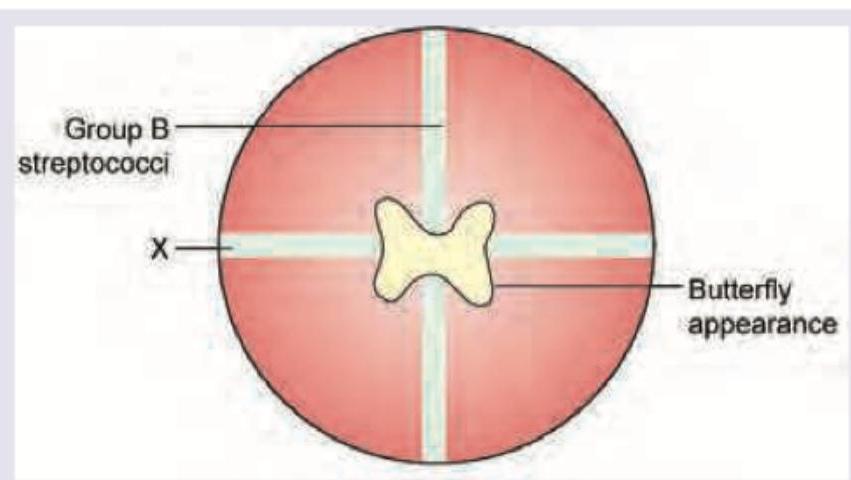
Which bacteria marked as $X$ is responsible for the pattern shown here in the culture plate?
The given image shows:
Practice by Chapter
Staphylococci
Practice Questions
Streptococci and Enterococci
Practice Questions
Neisseria and Moraxella
Practice Questions
Corynebacterium and Listeria
Practice Questions
Bacillus and Clostridium
Practice Questions
Enterobacteriaceae
Practice Questions
Vibrio, Aeromonas, and Plesiomonas
Practice Questions
Pseudomonas and Related Bacteria
Practice Questions
Haemophilus and HACEK Group
Practice Questions
Bordetella and Brucella
Practice Questions
Mycobacteria
Practice Questions
Spirochetes
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
