Infectious Diseases — MCQs

On this page
What is the most common cause of surgical wound infection?
Which statement is not true regarding Yaws?
Duffy blood group antigen negativity confers protection against infection by which Plasmodium species?
What is the most common cause of community-acquired pneumonia?
A 30-year-old pregnant woman develops severe sepsis with headache and neck stiffness after eating coleslaw, soft cheese, and cantaloupes at a potluck dinner. Two other women who attended the event develop self-limiting abdominal cramps, nausea, and vomiting. Which infectious agent is most likely responsible?
What is the empirical antibiotic treatment for a suspected case of pneumonia in an outpatient who has not received antibiotics in the past three months?
A dipstick kit was found to be positive. What is the treatment of choice?
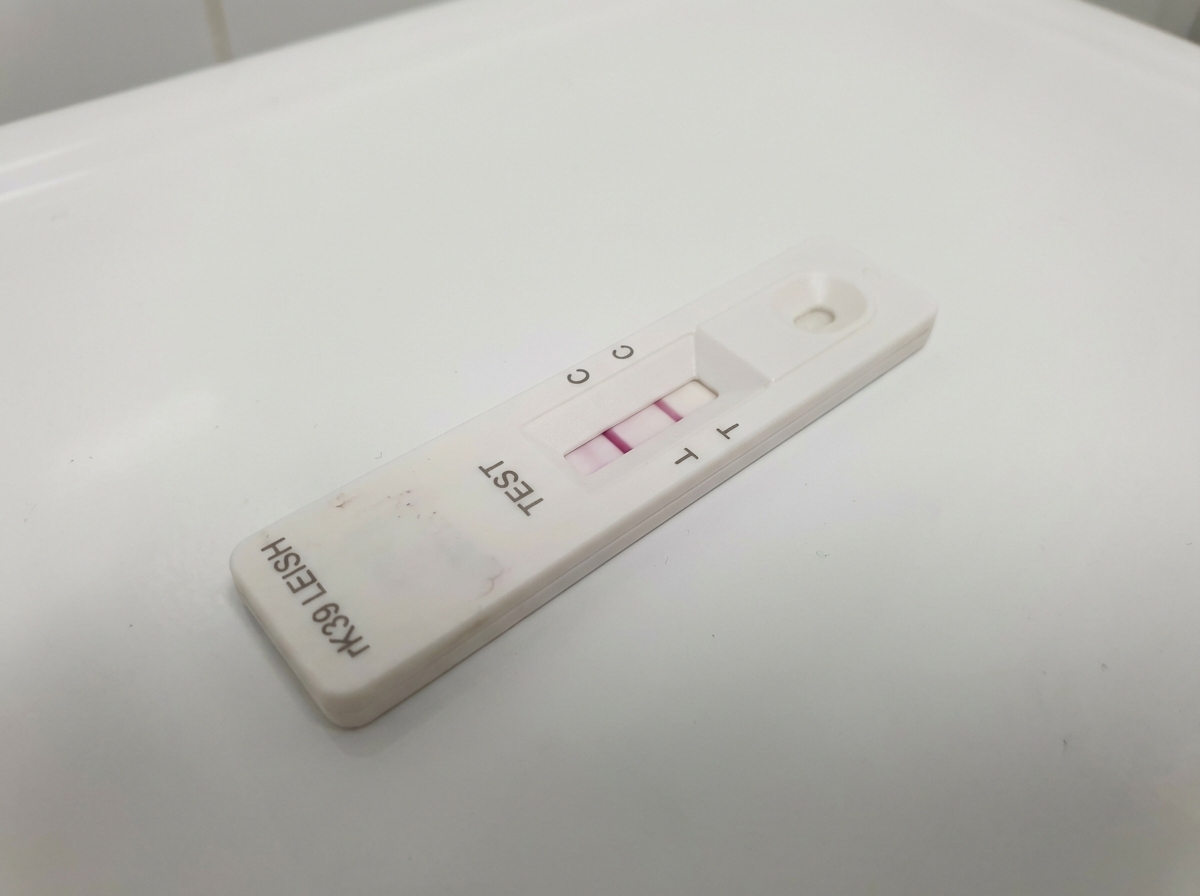
A farmer from Central Asia presents with an acute onset of swinging pyrexia, rigor, sweating, and monoarticular arthritis of the hip. He also complained of headache and insomnia. On examination, he is found to have small firm splenomegaly and hepatomegaly. His counts showed leucopenia with relative lymphocytosis. What is the most likely diagnosis?
Which of the following is NOT true about fluorescent antibody detection tests in the diagnosis of Plasmodium falciparum?
What is the most common diagnostic test for HIV infection?
Practice by Chapter
Principles of Antimicrobial Therapy
Practice Questions
Fever of Unknown Origin
Practice Questions
HIV/AIDS and Related Infections
Practice Questions
Tuberculosis and Mycobacterial Diseases
Practice Questions
Tropical and Parasitic Infections
Practice Questions
Viral Infections (Hepatitis, Herpes, etc.)
Practice Questions
Healthcare-Associated Infections
Practice Questions
Fungal Infections
Practice Questions
Sepsis and Septic Shock
Practice Questions
Infection in Immunocompromised Hosts
Practice Questions
Emerging and Re-emerging Infections
Practice Questions
Antimicrobial Resistance
Practice Questions
Vaccination Principles
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
