Enzymes — MCQs

On this page
Cyanides primarily inhibit which enzyme?
All of the following cells contain telomerase enzyme except?
If Km remains the same and Vmax is reduced, what type of enzyme inhibition is this?
Which enzyme is studied in the Triphenyl tetrazolium chloride test (TTC) for detecting myocardial infarction?
Which of the following enzymes does not utilize copper?
The enzyme phosphofructo kinase-1 is strongly activated by which of the following?
An increase in LDH-5 enzyme is seen in the following conditions, except:
Malonate is a competitive inhibitor of which of the following?
What is the enzyme involved in the following conversion?
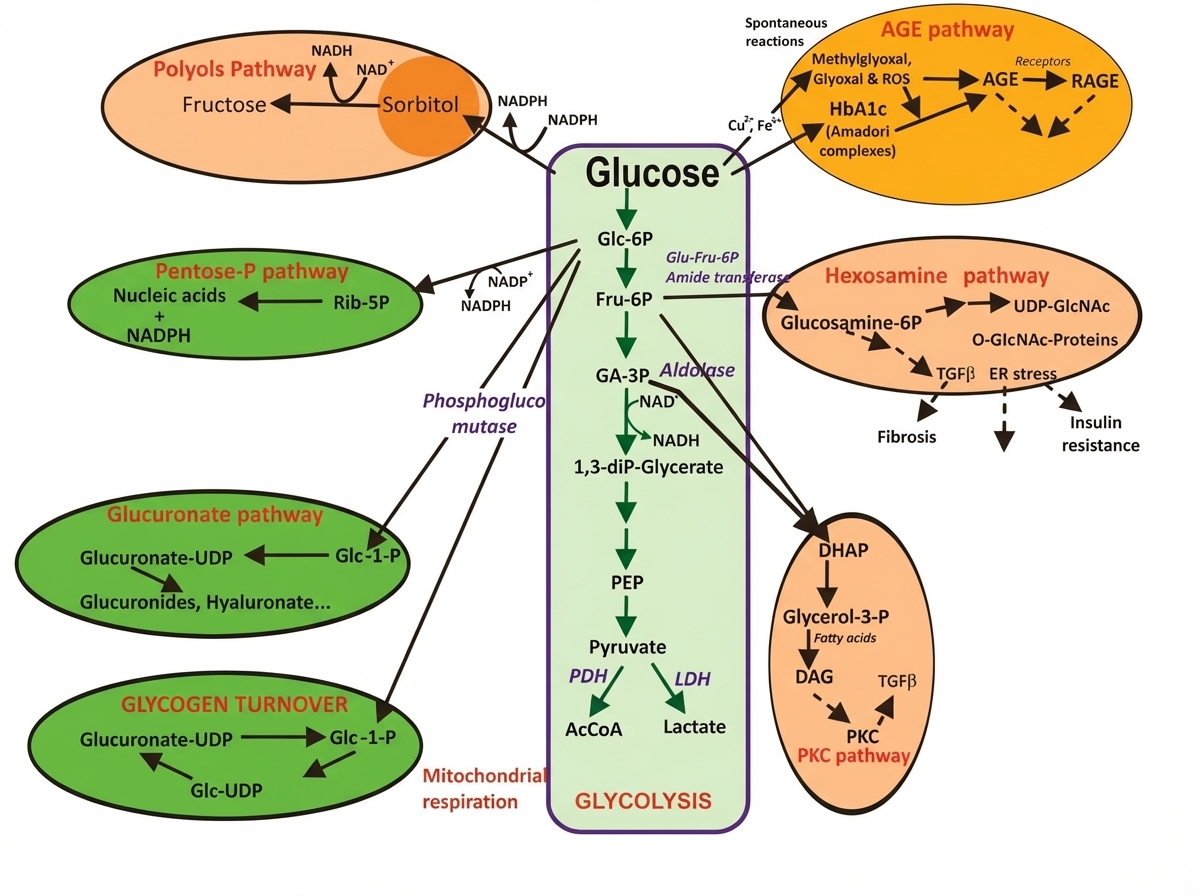
The enzyme trypsin is specific for peptide bonds of which type of amino acids?
Practice by Chapter
Enzyme Classification and Nomenclature
Practice Questions
Enzyme Kinetics and Michaelis-Menten Equation
Practice Questions
Enzyme Inhibition: Competitive and Non-competitive
Practice Questions
Allosteric Regulation
Practice Questions
Coenzymes and Cofactors
Practice Questions
Isoenzymes and Clinical Significance
Practice Questions
Enzyme Regulation: Covalent Modification
Practice Questions
Enzyme Regulation: Zymogen Activation
Practice Questions
Enzyme Induction and Repression
Practice Questions
Ribozymes and Catalytic RNA
Practice Questions
Enzyme Diagnostic Applications
Practice Questions
Enzyme Therapy and Inhibitors as Drugs
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
