Enzymes — MCQs

On this page
Which cellular component is responsible for providing cell shape and motility?
Which force does NOT act in an enzyme-substrate complex?
Trypsinogen is converted to trypsin by which of the following?
What is true about isoenzymes?
Which of the following is a non-competitive inhibitor of intestinal alkaline phosphatase?
What is the specific inhibitor of succinate dehydrogenase?
All of the following could include the mechanism or function of oxygenases, EXCEPT:
Identify the type of inhibition shown in the graph.
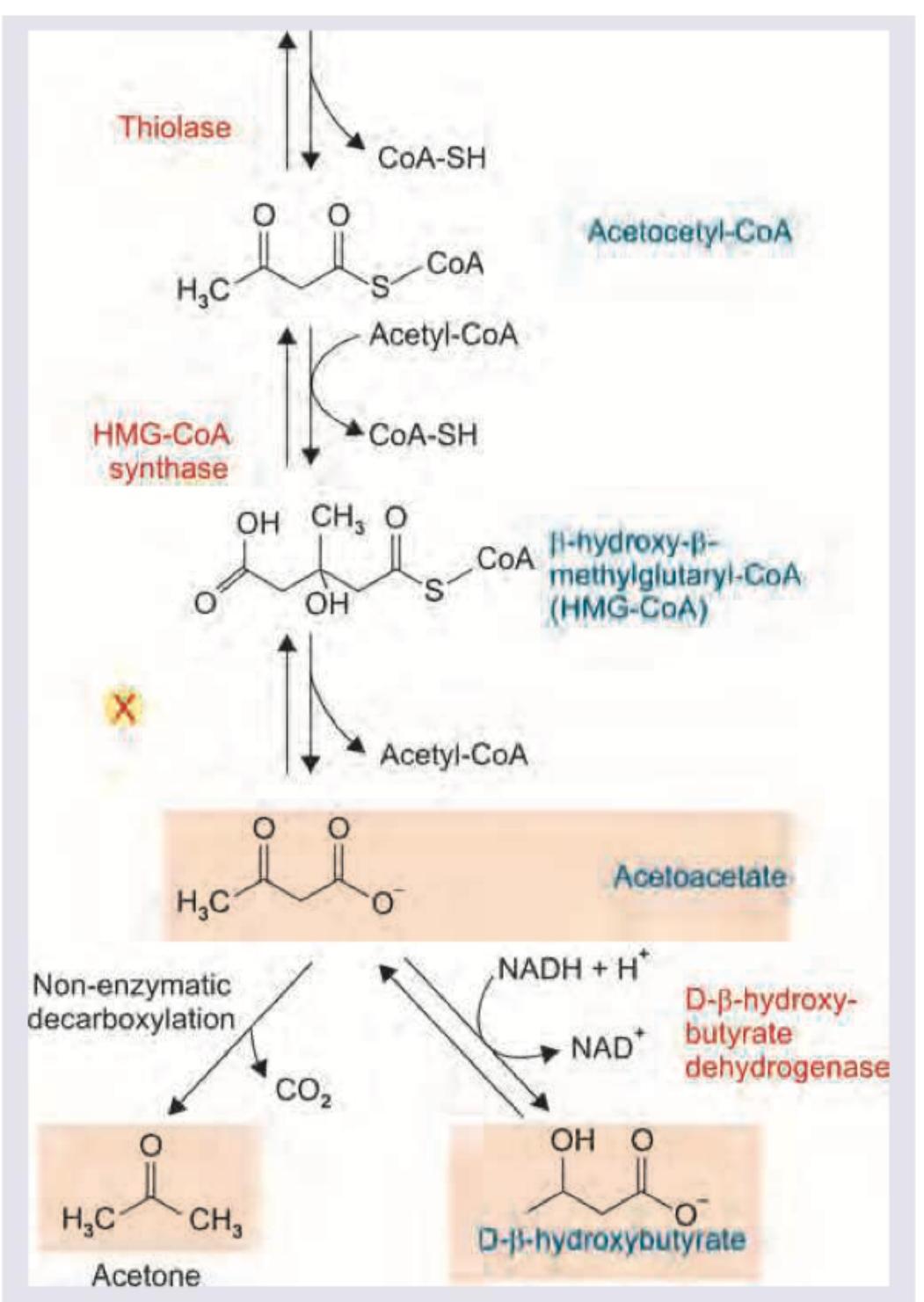
The image shows a biochemical pathway. Name the enzyme marked as X.
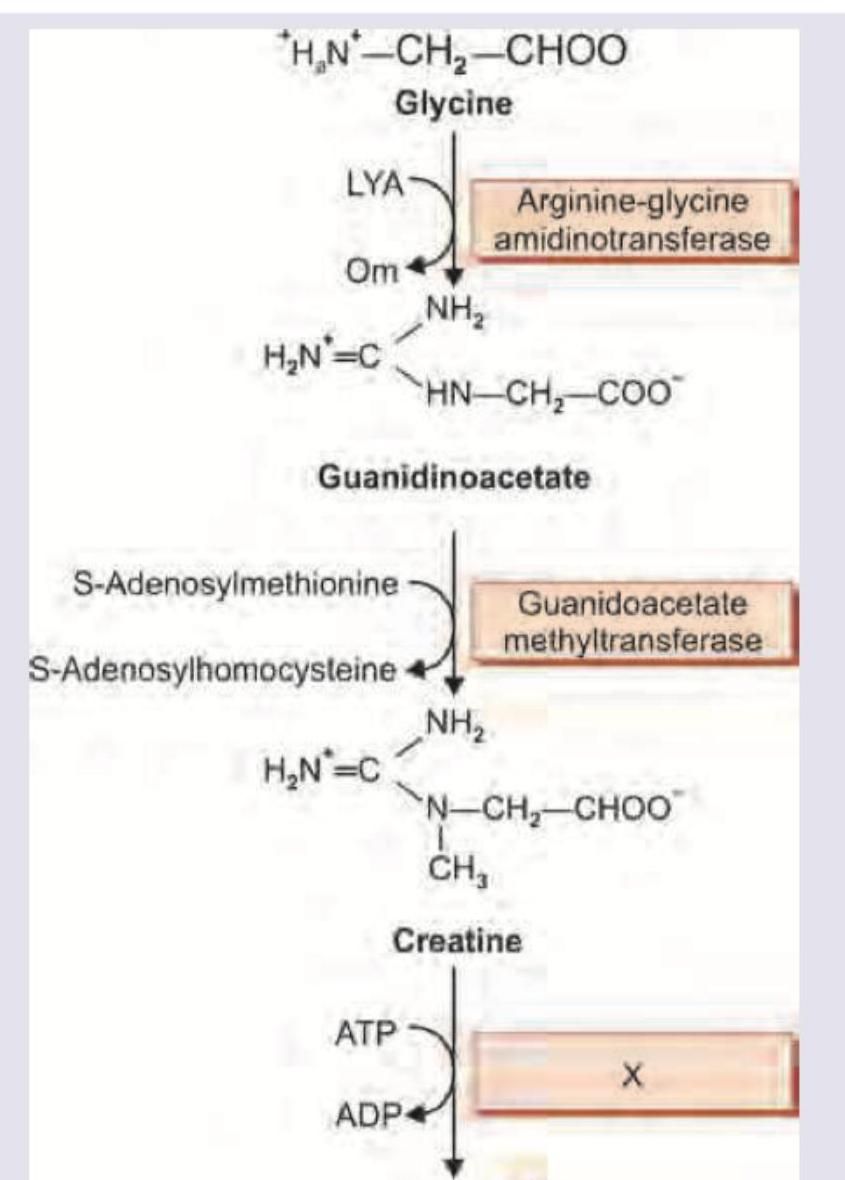
Name the enzyme marked as X in the reaction shown below.
Practice by Chapter
Enzyme Classification and Nomenclature
Practice Questions
Enzyme Kinetics and Michaelis-Menten Equation
Practice Questions
Enzyme Inhibition: Competitive and Non-competitive
Practice Questions
Allosteric Regulation
Practice Questions
Coenzymes and Cofactors
Practice Questions
Isoenzymes and Clinical Significance
Practice Questions
Enzyme Regulation: Covalent Modification
Practice Questions
Enzyme Regulation: Zymogen Activation
Practice Questions
Enzyme Induction and Repression
Practice Questions
Ribozymes and Catalytic RNA
Practice Questions
Enzyme Diagnostic Applications
Practice Questions
Enzyme Therapy and Inhibitors as Drugs
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
