Enzymes — MCQs

On this page
Which of the following is a competitive inhibitor of the succinate dehydrogenase enzyme?
Which of the following statements is FALSE regarding Cytochrome P-450 enzymes?
Which of the following is an example of a metalloenzyme?
Glutathione peroxidase is a/an?
Trypsin cleaves which amino acid residue?
Which of the following amino acids is a component of Thioredoxin reductase?
Regan enzyme is an isoenzyme of which of the following?
What is the action of adenylate cyclase?
For every 10-degree Celsius raise in temperature, the rate of most enzymatic reactions approximately:
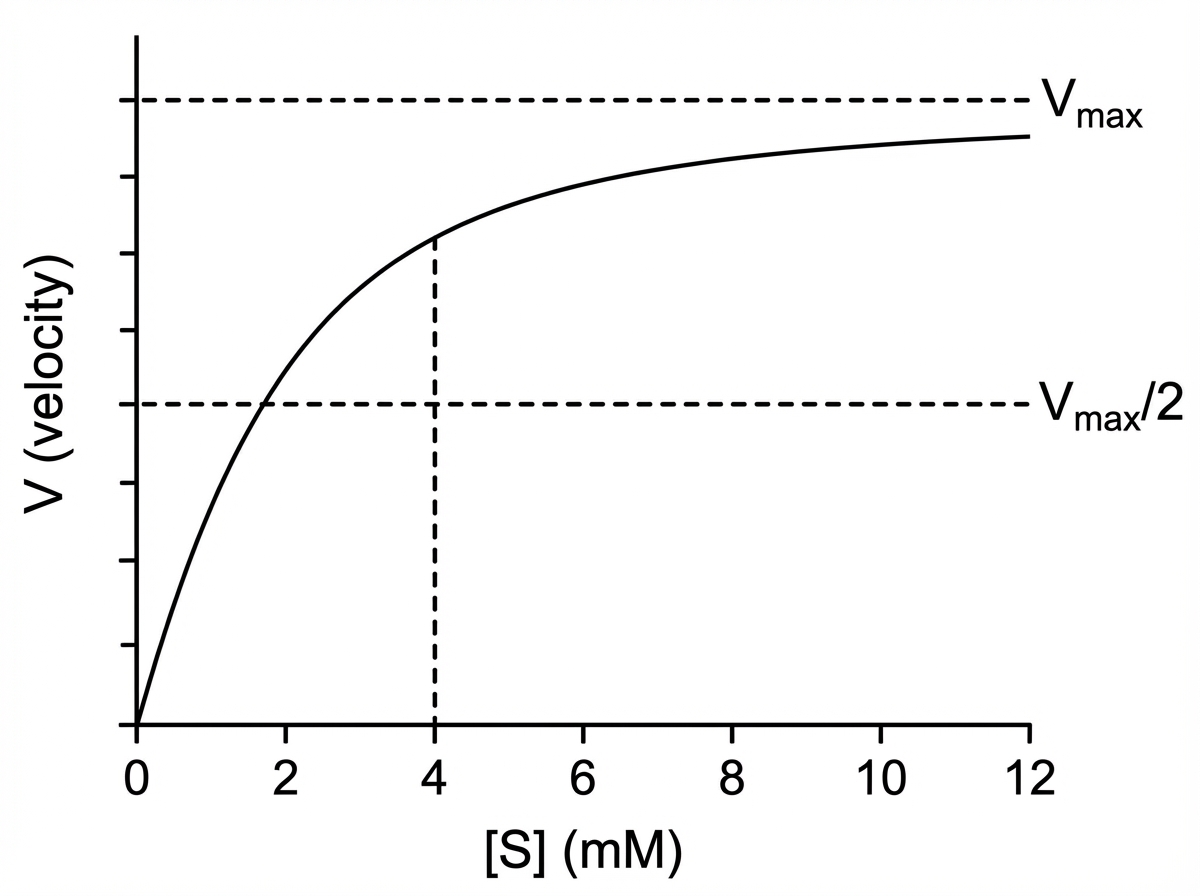
What is the Michaelis constant (Km)?
Practice by Chapter
Enzyme Classification and Nomenclature
Practice Questions
Enzyme Kinetics and Michaelis-Menten Equation
Practice Questions
Enzyme Inhibition: Competitive and Non-competitive
Practice Questions
Allosteric Regulation
Practice Questions
Coenzymes and Cofactors
Practice Questions
Isoenzymes and Clinical Significance
Practice Questions
Enzyme Regulation: Covalent Modification
Practice Questions
Enzyme Regulation: Zymogen Activation
Practice Questions
Enzyme Induction and Repression
Practice Questions
Ribozymes and Catalytic RNA
Practice Questions
Enzyme Diagnostic Applications
Practice Questions
Enzyme Therapy and Inhibitors as Drugs
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
