Enzymes — MCQs

On this page
Fumarase is classified as which type of enzyme?
Which of the following is true regarding Nitric Oxide Synthase?
NAD+ is reduced to NADH + H+ by dehydrogenases of all the following substrates, except?
What is the coenzyme for transamination reactions?
The conversion of glutamate to glutamine is catalysed by which type of enzyme?
Flipped pattern of LDH is seen in which of the following conditions?
Which enzyme is utilized by the Na/K pump?
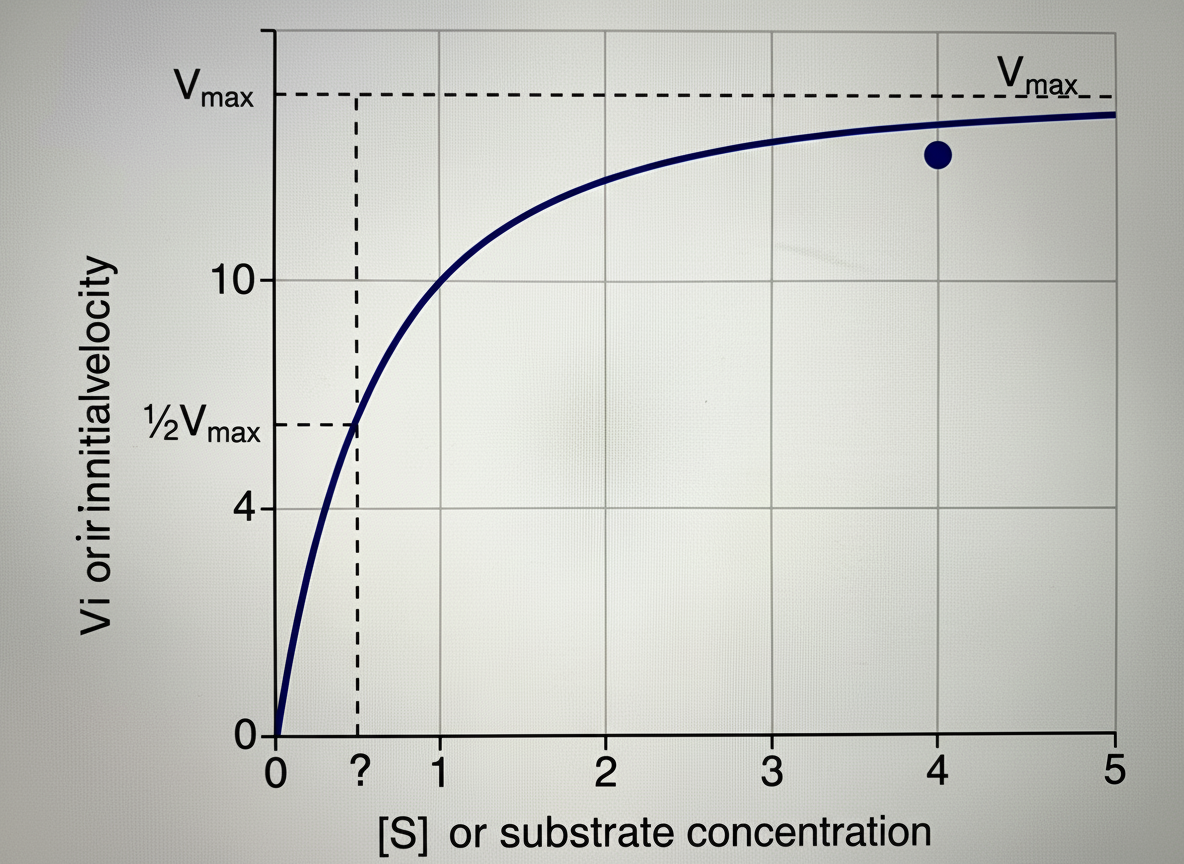
The provided graph illustrates the effect of substrate concentration on the initial velocity of an enzyme-catalyzed reaction. Identify the INCORRECT statement regarding this graph.
Which of the following statements is true about allosteric inhibition?
All of the following can be attributed to enzymatic catalysis, EXCEPT:
Practice by Chapter
Enzyme Classification and Nomenclature
Practice Questions
Enzyme Kinetics and Michaelis-Menten Equation
Practice Questions
Enzyme Inhibition: Competitive and Non-competitive
Practice Questions
Allosteric Regulation
Practice Questions
Coenzymes and Cofactors
Practice Questions
Isoenzymes and Clinical Significance
Practice Questions
Enzyme Regulation: Covalent Modification
Practice Questions
Enzyme Regulation: Zymogen Activation
Practice Questions
Enzyme Induction and Repression
Practice Questions
Ribozymes and Catalytic RNA
Practice Questions
Enzyme Diagnostic Applications
Practice Questions
Enzyme Therapy and Inhibitors as Drugs
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
