Enzymes — MCQs

On this page
Enzymes act by reducing which of the following?
Sulfonamides inhibit bacterial synthesis of folic acid by:
In myocardial infarction (MI), what happens to the ratio of Lactate Dehydrogenase (LDH) isoenzymes 1 and 2?
Allosteric enzymes show the following characteristics, except?
Which trace element is present in carbonic anhydrase?
Which enzyme is known as the 'suicidal enzyme'?
When the pH of a solution of a weak acid, HA, is equal to the pKa, what is the ratio of the concentrations of the conjugate base [A-] to the weak acid [HA]?
A patient presents with recurrent kidney stones. Microscopic examination of the urine specimen is shown below. Which of the following is not seen in the urine of this patient?
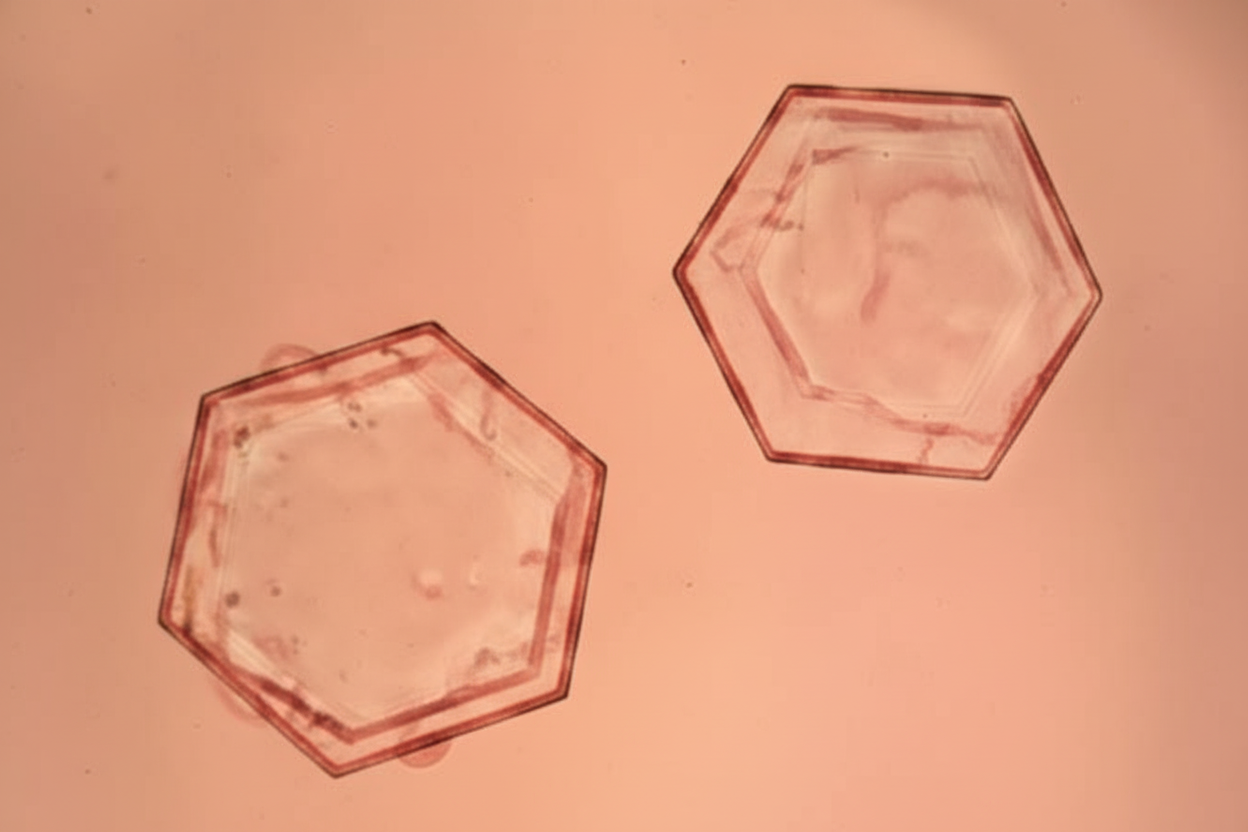
Arsenite inhibits which of the following enzymes?
Copper is a constituent of which enzyme?
Practice by Chapter
Enzyme Classification and Nomenclature
Practice Questions
Enzyme Kinetics and Michaelis-Menten Equation
Practice Questions
Enzyme Inhibition: Competitive and Non-competitive
Practice Questions
Allosteric Regulation
Practice Questions
Coenzymes and Cofactors
Practice Questions
Isoenzymes and Clinical Significance
Practice Questions
Enzyme Regulation: Covalent Modification
Practice Questions
Enzyme Regulation: Zymogen Activation
Practice Questions
Enzyme Induction and Repression
Practice Questions
Ribozymes and Catalytic RNA
Practice Questions
Enzyme Diagnostic Applications
Practice Questions
Enzyme Therapy and Inhibitors as Drugs
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
