Back
Covalent Modification of Enzymes: Biochemistry High-Yield Concepts for NEET PG and USMLE (2026)
Master covalent modification, phosphorylation, kinases, phosphatases, and zymogen activation for NEET PG and USMLE Step 1. High-yield enzyme regulation concepts with clinical correlations.

Covalent Modification of Enzymes: Biochemistry High-Yield Concepts for NEET PG and USMLE (2026)
You probably think enzyme regulation is all about allosteric changes and competitive inhibition. Wrong. The real game-changer in biochemistry is covalent modification — and if you dont master this concept, you'll miss 15-20% of enzyme regulation questions in NEET PG and USMLE Step 1.
Covalent modification controls everything from glycogen breakdown during exercise to protein digestion in your stomach. Unlike allosteric regulation that's reversible through simple binding and unbinding, covalent modifications actually change the enzyme's chemical structure. Some are reversible (like phosphorylation), others are permanent (like zymogen activation).
Here's what makes this concept absolutely critical: phosphorylation alone regulates over 30% of human proteins. When you see a question about glycogen phosphorylase, insulin signaling, or digestive enzymes, you're dealing with covalent modification. Master this, and you'll spot the patterns instantly.
What is Covalent Modification of Enzymes?
Covalent modification is the addition or removal of chemical groups to specific amino acid residues in enzymes through covalent bonds. Unlike the weak interactions in allosteric regulation, these modifications create permanent chemical changes that drastically alter enzyme activity.
The two main categories:
1. Reversible modifications: Can be added and removed as needed (phosphorylation, acetylation, methylation)
2. Irreversible modifications: Once done, cant be undone (zymogen activation, some acetylations)
This distinction is crucial because reversible modifications allow for dynamic regulation — turning enzymes on and off based on cellular needs — while irreversible modifications are used for one-time activations or permanent inactivations.
Phosphorylation: The Master Switch
Phosphorylation is the king of reversible covalent modifications. It involves adding phosphate groups (PO₄³⁻) to specific amino acids — mainly serine, threonine, and tyrosine residues.
Key players:
Kinases: Add phosphate groups using ATP
Phosphatases: Remove phosphate groups, releasing inorganic phosphate
The beauty of phosphorylation lies in its speed and reversibility. A kinase can phosphorylate an enzyme in milliseconds, completely changing its activity. A phosphatase can reverse this just as quickly.
High-yield exam fact: Phosphorylation can either activate OR inhibit enzymes. Dont memorize exceptions — understand the metabolic logic. For example:
Glycogen phosphorylase: Activated by phosphorylation (breaks down glycogen when energy is needed)
Acetyl-CoA carboxylase: Inhibited by phosphorylation (stops fatty acid synthesis when energy is low)
Practice identifying phosphorylation patterns with enzyme regulation questions and master the concepts through covalent modification lessons.
The Glycogen Phosphorylase Cascade: Exam Gold
This is the most tested example of covalent modification in both NEET PG and USMLE. You need to know it cold.
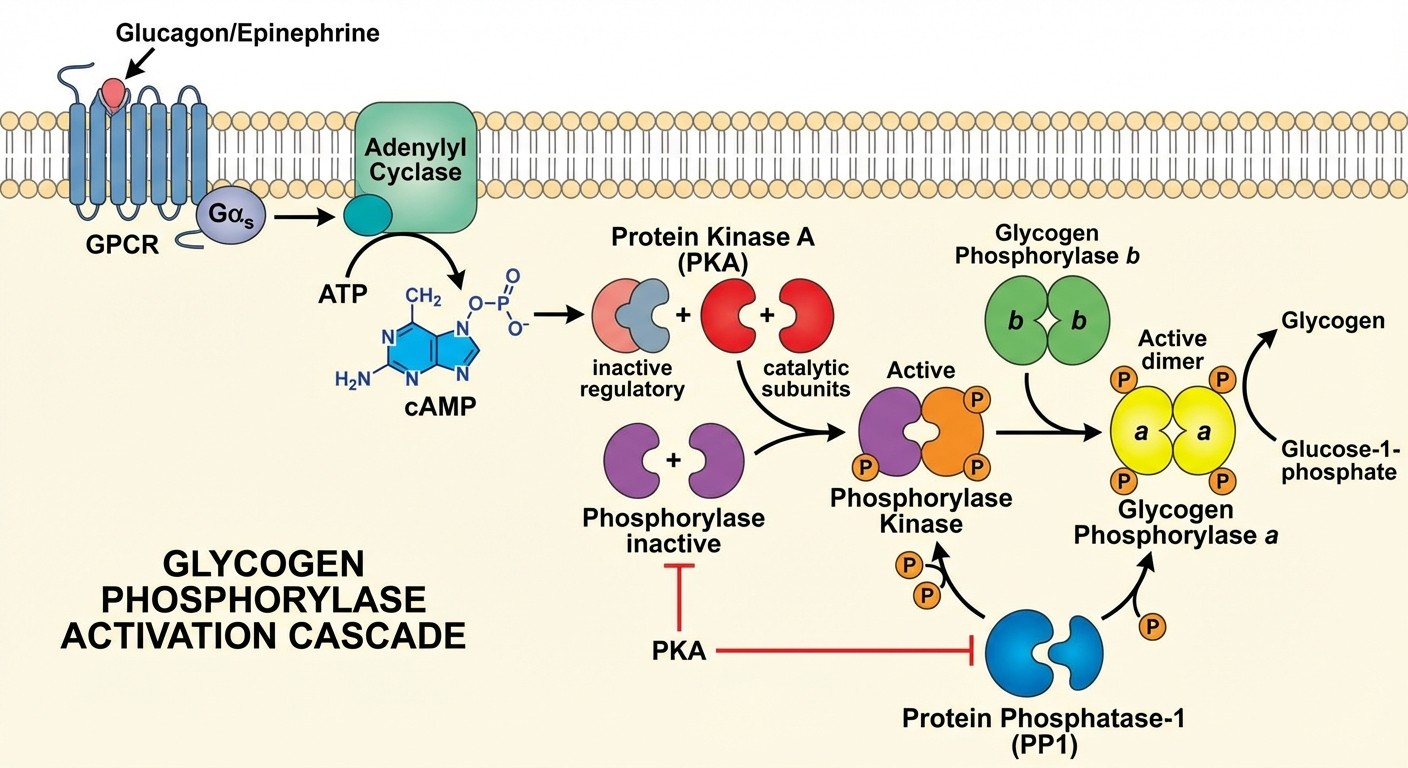
The cascade sequence: 1. Glucagon/Epinephrine binds to G-protein coupled receptors 2. Adenylyl cyclase increases cAMP levels 3. Protein kinase A (PKA) gets activated by cAMP 4. Phosphorylase kinase gets phosphorylated and activated by PKA 5. Glycogen phosphorylase b gets phosphorylated by phosphorylase kinase 6. Glycogen phosphorylase a (active form) breaks down glycogen The reversal:
Protein phosphatase-1 dephosphorylates both phosphorylase kinase and glycogen phosphorylase
This converts active phosphorylase a back to inactive phosphorylase b
Exam patterns to watch:
Questions testing the order of the cascade
Which step is rate-limiting (phosphorylase kinase activation)
How insulin opposes this pathway (activates phosphatases)
Clinical scenarios involving glycogen storage diseases
Reinforce this cascade with glycogen metabolism lessons and test your understanding with targeted flashcards.
Other Reversible Covalent Modifications
Adenylation (AMP Addition)
Best example: Glutamine synthetase in bacteria. Addition of AMP groups progressively inactivates the enzyme. While not heavily tested in medical exams, understanding this principle helps with enzyme regulation logic.
Acetylation
Histone acetylation is the classic example, though it's more relevant for gene regulation than metabolism. Acetyl groups are added to lysine residues. Exam relevance:
Generally associated with gene activation
Histone acetyltransferases (HATs) add acetyl groups
Histone deacetylases (HDACs) remove them
Methylation
Histone methylation can either activate or repress gene expression, depending on which lysine residue is methylated. Key points:
Unlike acetylation, methylation effects vary by location
Not easily reversible in the short term
More important for long-term gene regulation
Irreversible Covalent Modifications: Zymogen Activation
Zymogen activation is the irreversible conversion of inactive enzyme precursors (zymogens) into active enzymes through proteolytic cleavage. This is essential for digestive enzymes — you dont want proteases active inside the pancreas!
Chymotrypsinogen → Chymotrypsin
The classic example:
1. Chymotrypsinogen (inactive) is secreted by pancreatic acinar cells
2. Trypsin cleaves between Arg₁₅-Ile₁₆ in chymotrypsinogen
3. π-chymotrypsin (intermediate) is formed
4. α-chymotrypsin (fully active) results from further autolysis
Exam significance: This demonstrates how one active enzyme (trypsin) can activate multiple zymogens, creating an amplification cascade.
Pepsinogen → Pepsin
The gastric pathway:
1. Pepsinogen is secreted by chief cells
2. Low pH (1.5-2.0) in the stomach causes conformational change
3. Autocatalytic cleavage removes the inhibitory peptide
4. Pepsin becomes active and can activate more pepsinogen
High-yield fact: Pepsin is most active at pH 1.5-2.0, while chymotrypsin works optimally at pH 8.0-8.5. Questions often test optimal pH ranges for digestive enzymes.
Study zymogen activation patterns with protein digestion lessons and practice MCQs on zymogen activation.
Clinical Relevance: When Theory Meets Medicine
Understanding covalent modification isnt just academic — it explains how drugs work and diseases develop.
Aspirin and COX Enzymes
The mechanism: Aspirin irreversibly acetylates Ser₅³⁰ in cyclooxygenase-1 (COX-1) and Ser₅¹⁶ in cyclooxygenase-2 (COX-2). This permanently blocks the active site, preventing prostaglandin synthesis. Clinical significance:
Explains aspirin's long duration of action (until new COX enzymes are synthesized)
Why low-dose aspirin provides cardioprotection
Mechanism of aspirin-induced gastric irritation (COX-1 inhibition)
Insulin Resistance and Phosphorylation
Insulin resistance often involves disrupted phosphorylation cascades. When insulin receptor signaling is impaired, downstream kinases dont get properly activated, leading to poor glucose uptake.
Therapeutic Kinase Inhibitors
Many cancer drugs work by inhibiting specific kinases that are overactive in malignant cells. Understanding phosphorylation helps explain targeted therapy mechanisms.
High-Yield MCQ Patterns for NEET PG and USMLE
Pattern 1: Enzyme Activation vs Inactivation
Questions will give you a scenario and ask whether phosphorylation activates or inhibits the enzyme
Remember the metabolic logic: energy-producing enzymes are often activated by phosphorylation during stress
Pattern 2: Cascade Sequences
Expect questions testing the order of the glycogen phosphorylase cascade
Watch for "missing step" questions where you identify which enzyme comes next
Pattern 3: Clinical Correlations
Aspirin mechanism questions (irreversible acetylation)
Zymogen activation in pancreatic diseases
Kinase inhibitors in cancer therapy
Pattern 4: Regulation Comparison
Questions comparing allosteric vs covalent modification
Speed of regulation (covalent is faster than enzyme induction)
Reversibility patterns
Pattern 5: pH and Optimal Conditions
Pepsin vs chymotrypsin pH optima
Where zymogen activation occurs (stomach vs small intestine)
Master these patterns with enzyme regulation flashcards and test your knowledge with adaptive biochemistry questions.
How Oncourse AI Reinforces Covalent Modification Concepts
Oncourse AI's adaptive biochemistry module makes covalent modification stick through:
Spaced repetition timing: The AI identifies which modification types you struggle with (phosphorylation vs acetylation vs zymogen activation) and brings back challenging concepts right before you'd forget them. Pattern recognition training: Instead of just memorizing facts, Oncourse teaches you to spot exam patterns. When you see "glucagon stimulation," your brain automatically thinks "phosphorylation cascade." Clinical correlation integration: Every covalent modification concept is linked to real clinical scenarios. Aspirin becomes more than just "irreversible acetylation" — it becomes a complete understanding of how COX inhibition works. Adaptive difficulty progression: Start with basic phosphorylation, advance to complex cascades like glycogen phosphorylase, then tackle clinical applications. The AI ensures you master each level before progressing. Mistake pattern analysis: Oncourse tracks whether you confuse kinases with phosphatases, mix up zymogen activation sequences, or forget pH optima. It then creates personalized practice sets targeting your specific weak points.
The result? Instead of cramming hundreds of enzyme names, you understand the logic behind covalent modification and can predict enzyme behavior in any scenario.
Frequently Asked Questions
What is the difference between reversible and irreversible covalent modification?
Reversible modifications like phosphorylation can be undone by specific enzymes (phosphatases remove phosphate groups). Irreversible modifications like zymogen activation involve permanent structural changes — once chymotrypsinogen is cleaved to chymotrypsin, it cant be converted back.
How does phosphorylation change enzyme activity so dramatically?
Phosphate groups are large, negatively charged, and highly polar. Adding them to an enzyme can cause massive conformational changes, alter active site shape, affect substrate binding, or change protein-protein interactions. A single phosphorylation can switch an enzyme from completely inactive to fully active.
Why are digestive enzymes stored as inactive zymogens?
Proteases like chymotrypsin and pepsin would destroy pancreatic and gastric cells if they were active inside those tissues. Zymogen activation ensures these powerful enzymes only become active when they reach their target location (small intestine for chymotrypsin, stomach for pepsin).
Which amino acids can be phosphorylated?
Primarily serine, threonine, and tyrosine. Serine and threonine have hydroxyl (-OH) groups that can accept phosphate. Tyrosine phosphorylation is especially important in growth factor signaling and cancer biology.
How is the glycogen phosphorylase cascade regulated?
Multiple levels: glucagon/epinephrine activate it through cAMP and PKA, while insulin opposes it by activating protein phosphatase-1. AMP also allosterically activates phosphorylase b even without phosphorylation, providing an emergency glucose release mechanism.
What makes aspirin's effect on COX enzymes irreversible?
Aspirin forms a covalent bond with serine residues in the COX active site through acetylation. Unlike competitive inhibitors that bind and release, this covalent modification permanently blocks the enzyme. New COX enzymes must be synthesized to restore function.
Ready to master enzyme regulation through targeted practice? Prepare smarter with Oncourse AI — adaptive MCQs, spaced repetition, and AI explanations built for NEET PG and USMLE. Download free on Android and iOS.