Intro to Covalent Modification - Enzyme Makeovers
- Regulates enzyme activity by attaching/detaching chemical groups via covalent bonds.
- A key form of post-translational modification (PTM).
- Alters enzyme's conformation (3D structure) and/or active site.
- Leads to enzyme activation or inhibition.
- These modifications are enzyme-catalysed and typically reversible.
- Generally slower onset; effects are often more sustained/durable.
⭐ Covalent modification often leads to a more sustained regulatory effect compared to allosteric regulation, providing longer-term control over enzyme activity.
Phosphorylation/Dephosphorylation - Phospho-Play Power
- Phosphorylation: Covalent addition of phosphate ($PO_4^{3-}$) to specific amino acid residues.
- Catalyzed by Protein Kinases (e.g., Ser/Thr kinases, Tyr kinases).
- Reaction: $Protein + ATP \rightarrow Protein-PO_4 + ADP$.
- Dephosphorylation: Removal of $PO_4^{3-}$ from phosphoprotein.
- Catalyzed by Protein Phosphatases.
- Reaction: $Protein-PO_4 + H_2O \rightarrow Protein + P_i$.
- 📌 Mnemonic: Kinases 'Kindle' (add P), Phosphatases 'Pluck' (remove P).
- Mechanism: Alters protein conformation & thereby its activity (activation or inactivation).
- Key sites on protein: Hydroxyl groups of Serine (Ser), Threonine (Thr), Tyrosine (Tyr) residues.
- Significance: Most common, rapid, and reversible post-translational modification for enzyme regulation. Crucial in signal transduction pathways.

⭐ In glycogen metabolism, glycogen phosphorylase is activated by phosphorylation, while glycogen synthase is inactivated by phosphorylation, demonstrating reciprocal regulation.
Other Covalent Modifications - Tag Team Champs
- ADP-Ribosylation: $NAD^+$ is donor.
- Targets: Arg, Glu, Cys.
- Functions: DNA repair, signaling.
- Toxins: Cholera (↑Gsα), Pertussis (↓Giα), Diphtheria (↓eEF-2).
⭐ Diphtheria toxin catalyzes the ADP-ribosylation of elongation factor 2 (eEF-2), thereby inhibiting protein synthesis in eukaryotes.
- Methylation: SAM is donor.
- Targets: Histone Lys/Arg; DNA Cytosine.
- Function: Epigenetics.
- Acetylation: Acetyl-CoA is donor.
- Targets: Histone Lys.
- Function: Gene activation (HATs vs HDACs).
- Hydroxylation: Needs Vit C.
- Targets: Collagen Pro/Lys.
- Function: Collagen stability.
- Ubiquitination: Adds ubiquitin.
- Function: Proteasomal degradation, signaling.
- SUMOylation: Adds SUMO protein.
- Function: Nuclear processes, gene regulation.
- Function: Nuclear processes, gene regulation.
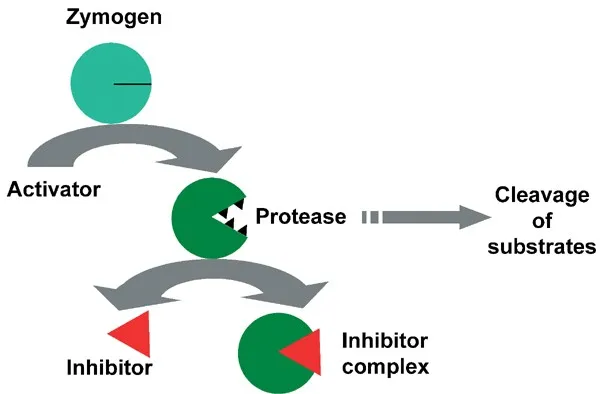
Proteolytic Cleavage (Zymogens) - The Big Snip!
- Zymogens (Proenzymes): Inactive enzyme precursors requiring proteolytic cleavage for activation. 📌 Zymogens are 'Lazy-gens' until activated.
- Mechanism: Irreversible covalent modification. Specific peptide bonds are cleaved, exposing the active site.
- A "one-way switch" - once activated, cannot be readily inactivated by reforming the bond.
- Significance:
- Prevents autodigestion (e.g., pancreas by its own proteases).
- Enables rapid enzyme deployment upon stimulus.
- Crucial for cascades (e.g., blood clotting, complement).
- Key Examples:
- Digestive Enzymes:
- Pepsinogen → Pepsin (stomach)
- Trypsinogen → Trypsin (pancreas)
- Chymotrypsinogen → Chymotrypsin (pancreas)
- Procarboxypeptidase → Carboxypeptidase (pancreas)
- Proelastase → Elastase (pancreas)
- Blood Clotting Factors: e.g., Prothrombin → Thrombin.
- Apoptosis: Procaspases → Caspases.
- Hormones: Proinsulin → Insulin.
- Digestive Enzymes:
⭐ Enteropeptidase (formerly enterokinase), secreted by the duodenal mucosa, is the key enzyme that initiates the activation of pancreatic zymogens by converting trypsinogen to active trypsin.
High‑Yield Points - ⚡ Biggest Takeaways
- Covalent modification: Reversible attachment/removal of chemical groups alters enzyme activity.
- Phosphorylation (by kinases) and dephosphorylation (by phosphatases) are most common.
- This changes enzyme conformation and thus its catalytic efficiency.
- Regulation speed: Slower than allosteric, but faster than enzyme synthesis/degradation.
- ATP typically serves as the phosphate group donor for kinases.
- Examples: Glycogen phosphorylase (activated by phosphorylation), pyruvate dehydrogenase (inactivated by phosphorylation).
- Other modifications: Adenylylation, ADP-ribosylation, methylation, acetylation.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

