Parasitology — MCQs

On this page
A 30-year-old female presents with abdominal pain, diarrhea, and weight loss. Stool microscopy reveals ova with a characteristic lateral spine. What is the most likely diagnosis?
A 15-year-old boy presented with fever and chills for 3 days. On examination, he was found to have delayed skin pinch time and dry oral mucosa. Identify the pathogen involved based on the provided peripheral blood smear image.
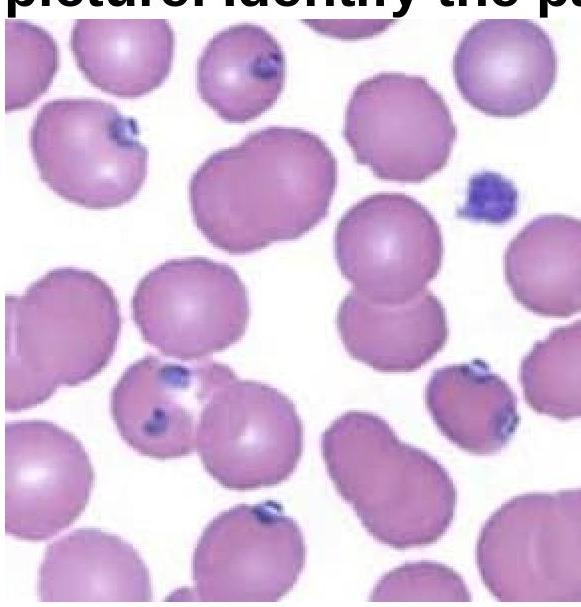
Identify the organism shown in the image.

What is a distinguishing feature of E. histolytica compared to E. dispar?
Promastigote form of Leishmania is found in which part of sandfly?
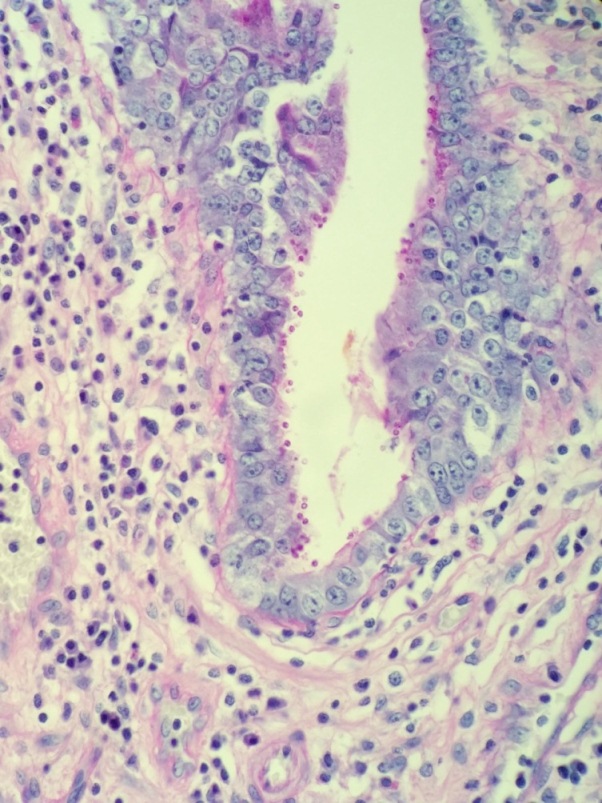
A 52-year-old male with HIV presents with profuse, watery diarrhea of 5 days’ duration. A biopsy of the small intestine is shown here. What is the most likely cause of this patient’s symptoms?
Calabar swelling is produced by?
How is diagnosis of filariasis confirmed most commonly?
Which stage of Leishmania is found in spleen aspirate of a patient?
Mucocutaneous leishmaniasis is caused by which of the following?
Practice by Chapter
Classification of Parasites
Practice Questions
Intestinal Protozoa
Practice Questions
Blood and Tissue Protozoa
Practice Questions
Malaria Parasites
Practice Questions
Leishmaniasis
Practice Questions
Intestinal Helminths: Nematodes
Practice Questions
Tissue Nematodes
Practice Questions
Trematodes
Practice Questions
Cestodes
Practice Questions
Ectoparasites
Practice Questions
Antiparasitic Drugs
Practice Questions
Laboratory Diagnosis of Parasitic Infections
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
