Nephrology — MCQs

On this page
What is the cause of hyperkalemia in chronic kidney failure?
In tubular necrosis, what is the ratio of urine to plasma creatinine?
Gitelman's Syndrome differs from Bartter's Syndrome in all except:
Dialysis disequilibrium syndrome is primarily caused by which of the following phenomena?
All of the following are used for the treatment of hyperkalemia except?
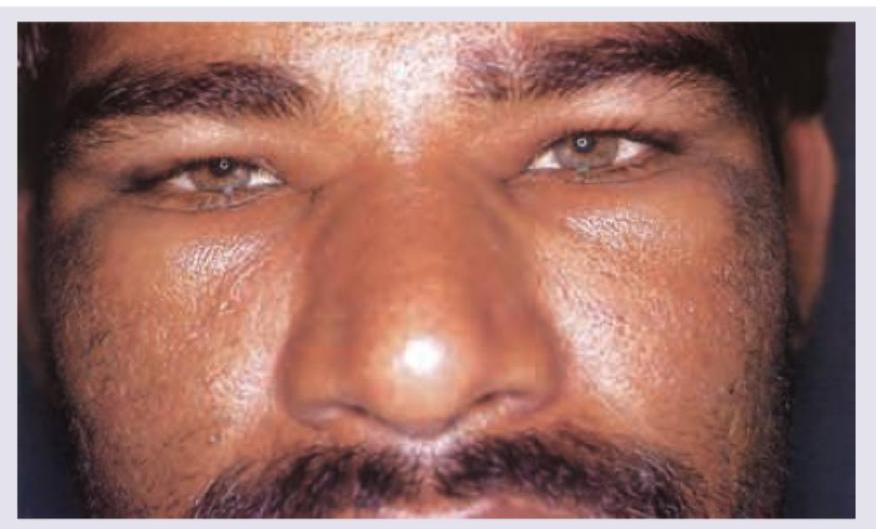
Which is the most common cause of this condition?
A 30-year-old man presents with generalized edema and hypertension. Urine examination shows subnephrotic proteinuria (< 2gm) and microscopic hematuria. Serum complement levels are decreased and he is positive for anti-hepatitis C antibodies. What is the most likely diagnosis?
The triad of hematuria, hypertension, and edema is a characteristic feature of which condition?
Presence of which of the following in the urine is diagnostic of glomerular injury?
Which of the following conditions is typically associated with hyperphosphatemia?
Practice by Chapter
Acute Kidney Injury
Practice Questions
Chronic Kidney Disease
Practice Questions
Glomerular Diseases
Practice Questions
Tubulointerstitial Diseases
Practice Questions
Nephrotic and Nephritic Syndromes
Practice Questions
Urinary Tract Infections
Practice Questions
Renal Replacement Therapy
Practice Questions
Fluid and Electrolyte Disorders
Practice Questions
Acid-Base Disorders
Practice Questions
Kidney in Systemic Diseases
Practice Questions
Kidney Stones and Obstructive Uropathy
Practice Questions
Hypertension in Kidney Disease
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
