Infectious Diseases — MCQs

On this page
Which of the following organisms would typically be found in a patient with atypical community-acquired pneumonia?
A 40-year-old woman, found to be positive for HBsAg, is excluded from blood donation due to a chronic shortage of blood. She is asymptomatic with normal liver function tests and negative serologic tests for IgM anti-HAV, anti-HBc, and anti-HCV. Repeat testing 6 months later shows the same results. Which of the following is the most appropriate statement regarding the pathophysiology of this patient's condition?
A 33-year-old woman presented with a 1-day history of fever and chills. On examination, she was febrile with a blood pressure of 70/40 mmHg. Over several hours, a widespread erythrodermic rash developed, and she collapsed. Further questioning revealed that the patient had removed a tampon shortly before presentation as she had just ceased menstruating. What is the most likely diagnosis?
A patient with AIDS is receiving therapy with Zidovudine, Lamivudine, and Indinavir. The patient develops pulmonary tuberculosis and requires treatment initiation. Which of the following medications should be avoided in this patient?
Which of the following is NOT a characteristic of secondary syphilis?
A young male admitted for pneumonia for 5 days develops gripping abdominal pain and loose stools. Which drug(s) might be beneficial in managing these new symptoms?
A 30-year-old patient presented with a 30-day history of jaundice. Liver function tests showed a bilirubin of 100mg/dL, SGOT/SGPT of 100/1450, and serum alkaline phosphatase of 240 IU. The patient was positive for HBsAg. What is the confirmatory test to establish acute Hepatitis B infection?
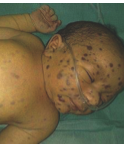
What is the diagnosis in a baby girl delivered at 30 weeks' gestation?
What is the diagnostic test of choice for neurosyphilis?
Most West Africans and people with origins in that region carry the Duffy-negative FyFy phenotype and are therefore resistant to______ malaria?
Practice by Chapter
Principles of Antimicrobial Therapy
Practice Questions
Fever of Unknown Origin
Practice Questions
HIV/AIDS and Related Infections
Practice Questions
Tuberculosis and Mycobacterial Diseases
Practice Questions
Tropical and Parasitic Infections
Practice Questions
Viral Infections (Hepatitis, Herpes, etc.)
Practice Questions
Healthcare-Associated Infections
Practice Questions
Fungal Infections
Practice Questions
Sepsis and Septic Shock
Practice Questions
Infection in Immunocompromised Hosts
Practice Questions
Emerging and Re-emerging Infections
Practice Questions
Antimicrobial Resistance
Practice Questions
Vaccination Principles
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
