Cardiology — MCQs

On this page
A 60-year-old female presented to the ER with chest pain and dyspnea, along with palpitations, nausea, vomiting, and a few episodes of syncope. MI was suspected. ECG and lab findings revealed raised cardiac enzymes. ECHO was also done. The patient was planned for coronary angiography, but no coronary artery stenosis was observed. What is the drug of choice for the above condition?
Which of the following arteries is most common to undergo atherosclerotic changes leading to coronary artery disease?
Which of the following conditions is characterized by an early diastolic murmur?
Water hammer pulse is also known as:
All of the following factors predispose to Aortic dissection, EXCEPT:
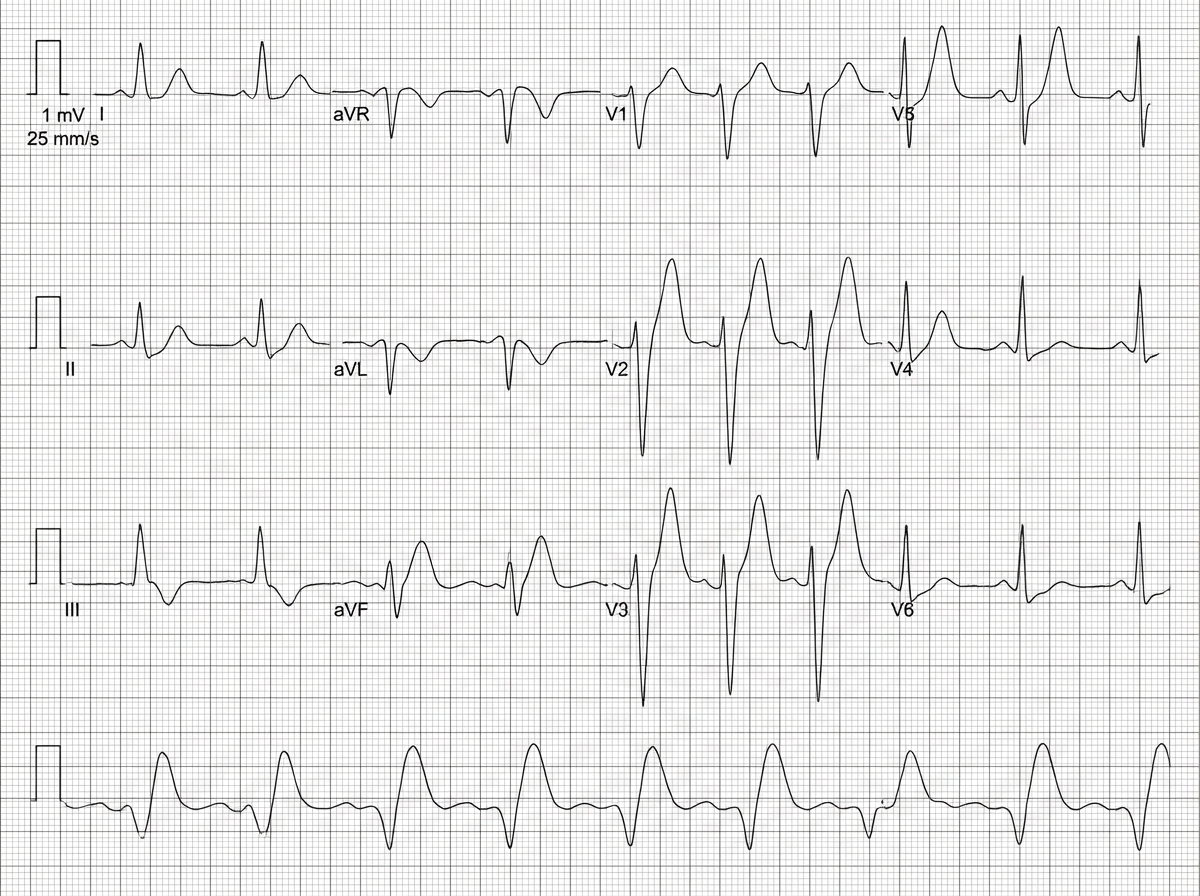
A patient presents to the emergency department in an unconscious state after a 15-minute delay following a call. The patient has a barely palpable radial pulse and no signs of trauma. The systolic blood pressure is 58 mm Hg. An ECG and ultrasound film are shown. Which electrolyte abnormality is most likely to cause this clinical scenario?
Low renin hypertension is seen in all of the following conditions, except:
Acute aortic regurgitation is seen in all the following conditions except?
In Myocardial infarction, what is the most specific marker?
What is true about pulsus paradoxus?
Practice by Chapter
Coronary Artery Disease and Angina
Practice Questions
Acute Coronary Syndromes
Practice Questions
Heart Failure
Practice Questions
Cardiac Arrhythmias
Practice Questions
Valvular Heart Diseases
Practice Questions
Cardiomyopathies
Practice Questions
Pericardial Diseases
Practice Questions
Congenital Heart Disease in Adults
Practice Questions
Hypertension and Hypertensive Emergencies
Practice Questions
Pulmonary Hypertension
Practice Questions
Non-invasive Cardiac Diagnostics
Practice Questions
Preventive Cardiology
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
