Cardiology — MCQs

On this page
Bundle of Kent is seen in which of the following conditions?
Which type of myocardial infarction is most likely to cause fatal arrhythmias?
A young man, previously diagnosed with congenital heart disease with a left-to-right shunt, has a very loud pansystolic murmur at the left sternal edge without any hemodynamic disturbance. What is the most likely diagnosis?
All of the following are signs of pulmonary hypertension, except?
Renal artery stenosis may occur in all of the following except?
The Stanford classification is used to represent which of the following?
Reperfusion is useful for which of the following types of myocardium?
All of the following are true about Atrial Septal Defect (ASD) except?
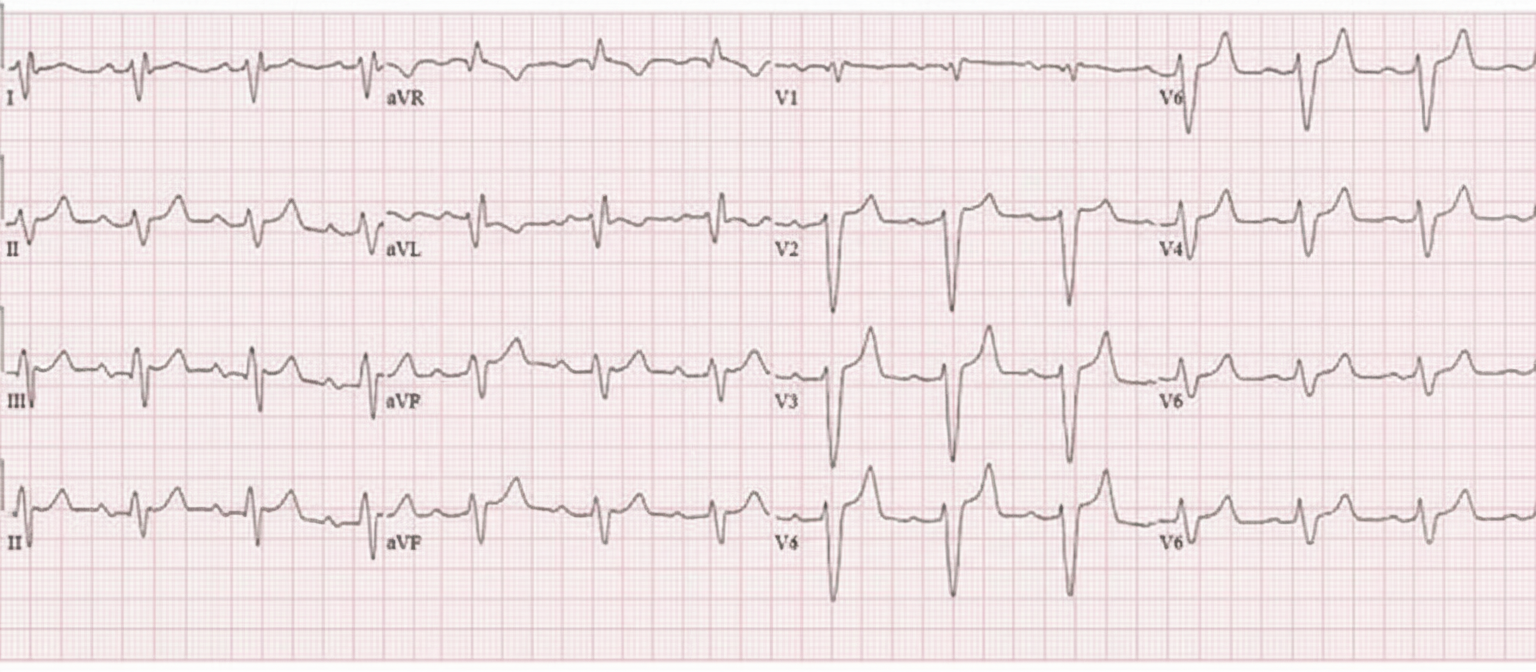
A 50-year-old female patient on losartan and spironolactone therapy for hypertension developed hypotension and bradycardia. The ECG taken is as shown. What is the effective treatment for her condition?
Pulsus alternans is seen in which of the following conditions?
Practice by Chapter
Coronary Artery Disease and Angina
Practice Questions
Acute Coronary Syndromes
Practice Questions
Heart Failure
Practice Questions
Cardiac Arrhythmias
Practice Questions
Valvular Heart Diseases
Practice Questions
Cardiomyopathies
Practice Questions
Pericardial Diseases
Practice Questions
Congenital Heart Disease in Adults
Practice Questions
Hypertension and Hypertensive Emergencies
Practice Questions
Pulmonary Hypertension
Practice Questions
Non-invasive Cardiac Diagnostics
Practice Questions
Preventive Cardiology
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
