Cardiology — MCQs

On this page
Which gene alteration is most commonly associated with dilated cardiomyopathy?
Comment on the diagnosis of the patient based on ECG?
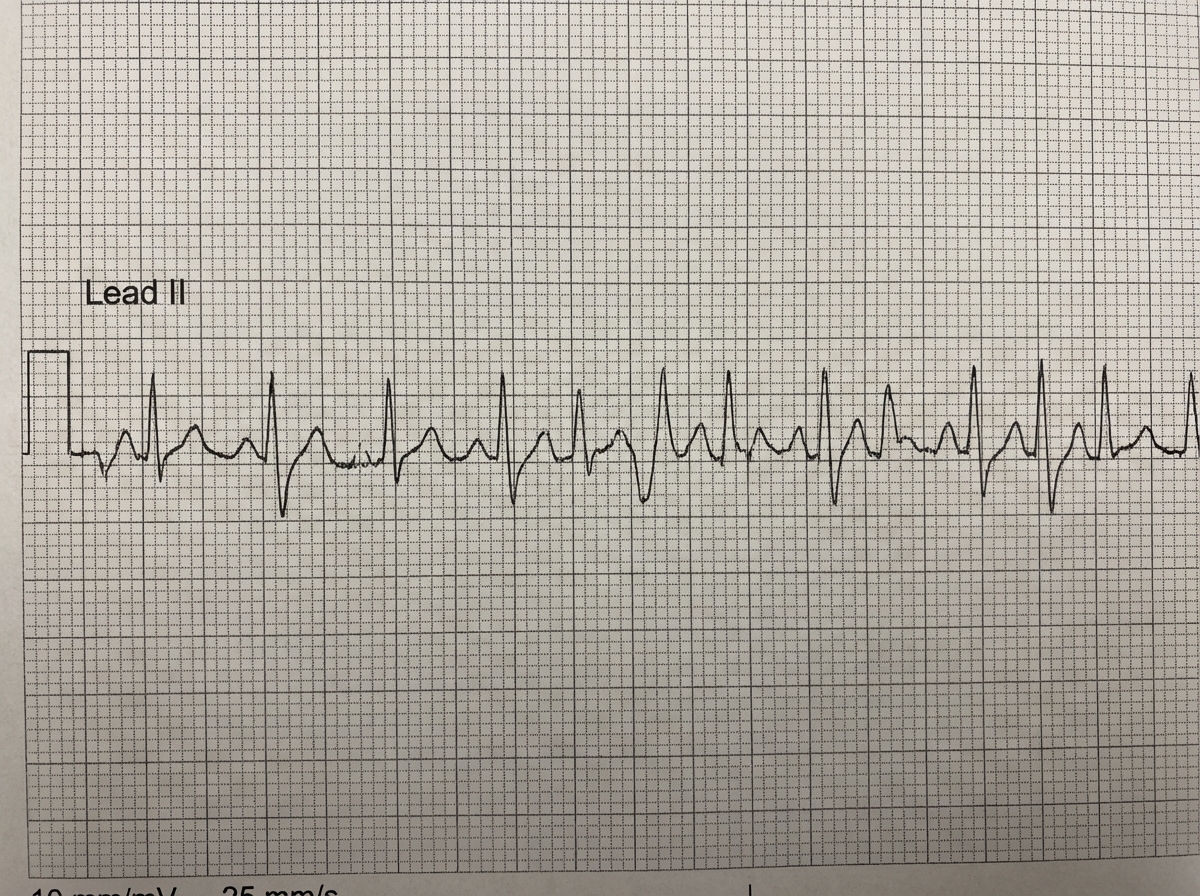
What is the most common cause of arrhythmia?
Which infection is known to cause Coronary Artery Disease (CAD)?
A fixed PR interval with occasional dropped beats in a 2:1, 3:1, or 4:1 pattern, accompanied by a wide QRS complex, is characteristic of which condition?
What is the most common cause of embolism leading to cerebrovascular disease?
A 45-year-old man presents to the emergency department with dizziness. He has a history of supraventricular beats and is currently taking aspirin, atenolol, and quinine. His EKG reveals Torsades De Pointes. How should he be managed?
A 50-year-old man has a 2-year history of angina pectoris that occurs during exercise. On physical examination, his blood pressure is 135/75 mm Hg, and his heart rate is 79/min and slightly irregular. Coronary angiography shows a fixed 75% narrowing of the anterior descending branch of the left coronary artery. He has several risk factors for atherosclerosis: smoking, hypertension, and hypercholesterolemia. Which of the following is the earliest event resulting from the effects of these factors?
Severity of mitral stenosis is judged by?
Electrical alternans is seen in which of the following conditions?
Practice by Chapter
Coronary Artery Disease and Angina
Practice Questions
Acute Coronary Syndromes
Practice Questions
Heart Failure
Practice Questions
Cardiac Arrhythmias
Practice Questions
Valvular Heart Diseases
Practice Questions
Cardiomyopathies
Practice Questions
Pericardial Diseases
Practice Questions
Congenital Heart Disease in Adults
Practice Questions
Hypertension and Hypertensive Emergencies
Practice Questions
Pulmonary Hypertension
Practice Questions
Non-invasive Cardiac Diagnostics
Practice Questions
Preventive Cardiology
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
