Carbohydrate Metabolism — MCQs

On this page
Which of the following enzymes does NOT increase its activity during starvation?
Von Gierke's disease occurs due to deficiency of which enzyme?
Arrange the following 4 enzymes of gluconeogenesis in sequence:
The development of cataracts in patients with Diabetes Mellitus is primarily due to the accumulation of which of the following substances in the lens?
A patient with a history of strenuous exercise skipped a meal and later consumed alcohol at a party. Alcohol inhibits which of the following biochemical processes, potentially leading to hypoglycemia?
A patient with a long-standing history of diabetes presents with cataracts. Which of the following metabolic products is primarily responsible for cataract formation in this condition?
A 68-year-old female patient has had a diabetic cataract for 3 months. Accumulation of which of the following substances is responsible for this?
Which of the following cell pathways occurs exclusively in cell cytoplasm?
Cancer cells take up excess glucose because?
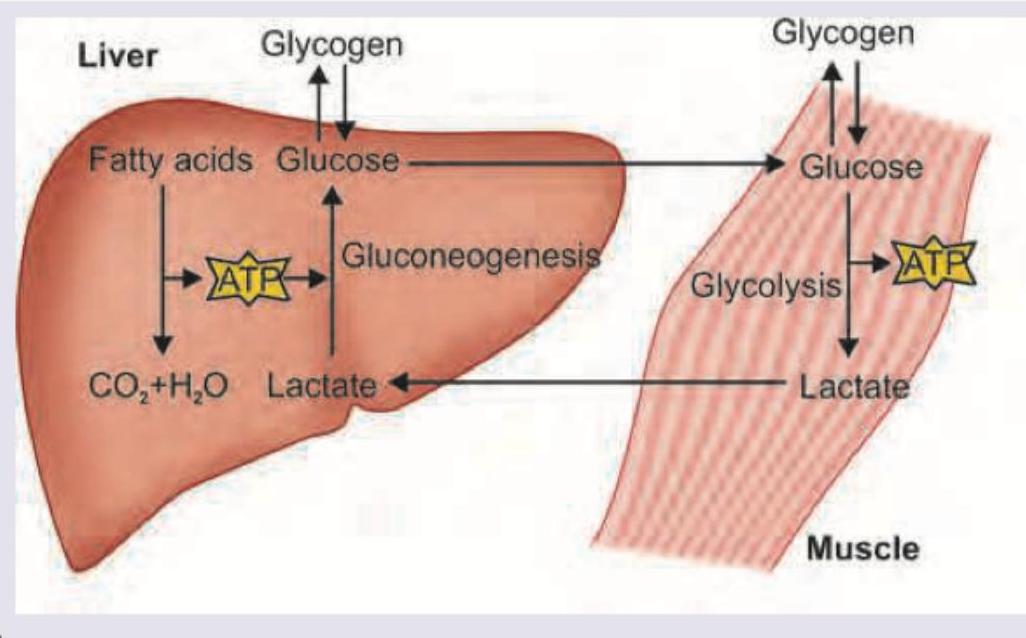
What is the cycle shown below called?
Practice by Chapter
Carbohydrate Chemistry and Classification
Practice Questions
Glycolysis: Reactions and Regulation
Practice Questions
Gluconeogenesis: Reactions and Regulation
Practice Questions
Glycogen Metabolism: Synthesis and Breakdown
Practice Questions
Glycogen Storage Diseases
Practice Questions
Pentose Phosphate Pathway
Practice Questions
Metabolism of Fructose and Galactose
Practice Questions
Disorders of Fructose and Galactose Metabolism
Practice Questions
Blood Glucose Regulation
Practice Questions
Diabetes Mellitus: Biochemical Aspects
Practice Questions
Glycosylation and Glycoproteins
Practice Questions
Lactose Intolerance and Galactosemia
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
