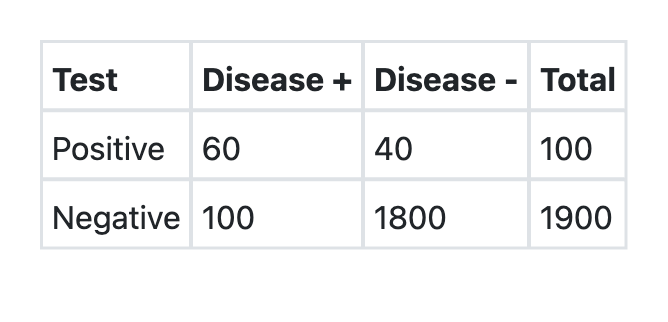
A new test was developed for detection of COVID-19. What is the sensitivity of the test as per the information provided above?
A study was conducted to evaluate the effectiveness of a new antidiabetic drug. The fasting blood glucose levels (mg/dL) of 5 diabetic patients after 3 months of treatment were: 110, 94, 102, 98, 96. Consider the following statements about this data: 1. The range of blood glucose levels is 16 mg/dL 2. The median blood glucose level is 98 mg/dL 3. The standard deviation is √10 mg/dL
Which of the following are correct in respect of Diphtheria? 1. The incubation period is 14 to 28 days 2. Diphtheria antitoxin is used in treatment of cases 3. It is one of the diseases protected from, by the Pentavalent vaccine given in National Program
A person has presented with history of dog-bite on the uncovered surface of his right leg. There is a minor abrasion without bleeding. Consider the following statements about management : 1. The bite should be taken as a category I contact with suspect animal 2. The bite requires local treatment of the wound 3. The person requires immediate vaccination 4. The person requires immediate administration of rabies immunoglobulin Which of the above statements are correct ?
Consider the following stages of modern sewage treatment plants : 1. Use of Grit chamber 2. Use of Primary sedimentation tank 3. Screening 4. Use of Sludge digester What would be the correct sequence of the above stages ?
A vital layer, also known as "Schmutzdecke" is seen in which one of the following ?