Mycology

On this page
🔬 Fungal Kingdom Mastery: The Hidden Microbial Empire
Fungi occupy a unique evolutionary niche between plants and animals, wielding remarkable abilities to shift forms, evade immune defenses, and resist therapeutic attack. You'll master how temperature triggers dimorphic transformations, recognize diagnostic morphological patterns that distinguish species, understand the molecular battleground between host and pathogen, and deploy targeted antifungal strategies while navigating emerging resistance threats. This lesson builds a systematic framework for rapid clinical decision-making in mycological infections, transforming abstract microbiology into actionable diagnostic and therapeutic precision.
📌 Remember: YEAST-MOLD-DIMORPHIC - Yeasts are unicellular (37°C favored), Molds are multicellular filamentous (25°C favored), Dimorphic switch between forms based on temperature and environment
Fungal infections affect >1.5 billion people globally, with invasive mycoses causing >1.5 million deaths annually. Immunocompromised patients face 30-90% mortality rates from invasive fungal infections, making rapid diagnosis and appropriate therapy critical for survival.
-
Fungal Cell Structure Mastery
- Cell wall contains chitin and β-glucans (absent in bacteria)
- Ergosterol replaces cholesterol in membrane (key antifungal target)
- Nucleus is membrane-bound with linear chromosomes
- DNA content: 10-100 megabases per genome
- Gene density: 2,000-20,000 genes per species
-
Reproductive Strategies
- Asexual reproduction: Conidia, budding, fragmentation
- Sexual reproduction: Ascospores, basidiospores, zygospores
- Parasexual cycle: Genetic recombination without meiosis
- Occurs in Aspergillus and Candida species
- Enables antifungal resistance development
⭐ Clinical Pearl: Fungal infections show biphasic growth patterns - most pathogenic fungi grow as molds at 25°C (environmental) and yeasts at 37°C (human body temperature), explaining their successful human adaptation
| Fungal Type | Temperature | Morphology | Reproduction | Clinical Significance | Examples |
|---|---|---|---|---|---|
| Yeasts | 37°C optimal | Unicellular, budding | Budding, fission | Bloodstream infections | Candida, Cryptococcus |
| Molds | 25°C optimal | Multicellular, hyphae | Conidia, spores | Pulmonary infections | Aspergillus, Mucor |
| Dimorphic | Both temps | Temperature-dependent | Both mechanisms | Systemic mycoses | Histoplasma, Blastomyces |
| Dermatophytes | 25-30°C | Septate hyphae | Macro/microconidia | Skin/nail infections | Trichophyton, Microsporum |
| Zygomycetes | 37°C rapid | Nonseptate hyphae | Sporangiospores | Rhinocerebral disease | Rhizopus, Mucor |
Understanding fungal classification systems enables systematic approach to diagnosis, where morphological characteristics combined with biochemical testing and molecular methods provide definitive species identification essential for targeted antifungal therapy selection.
🔬 Fungal Kingdom Mastery: The Hidden Microbial Empire
🌡️ Temperature-Driven Transformation: The Dimorphic Switch
📌 Remember: HBCP-MOLD-YEAST - Histoplasma, Blastomyces, Coccidioides, Paracoccidioides exist as MOLD in environment (25°C) and YEAST in tissue (37°C), except Coccidioides forms spherules
The dimorphic transition involves complex gene regulation cascades triggered by temperature, CO₂ concentration, and nutrient availability. This switch requires 24-72 hours for complete morphological transformation and involves differential protein expression of over 1,000 genes.
-
Molecular Switch Mechanisms
- Heat shock proteins (HSP90, HSP70) regulate transformation
- cAMP-dependent pathways control morphogenesis
- Transcription factors alter gene expression profiles
- >50% of genome shows differential expression
- Switch completes within 48-96 hours
- Irreversible once established in tissue
-
Clinical Transformation Patterns
- Histoplasma: Mold → 2-4 μm intracellular yeasts
- Blastomyces: Mold → 8-15 μm broad-based budding yeasts
- Coccidioides: Mold → 20-200 μm spherules with endospores
- Spherules contain 200-300 endospores
- Endospore release triggers intense inflammation
⭐ Clinical Pearl: Dimorphic fungi cause >90% of endemic mycoses in immunocompetent hosts, with geographic distribution patterns reflecting optimal environmental growth conditions and specific ecological niches
| Organism | Environmental Form | Tissue Form | Size (μm) | Geographic Distribution | Mortality Rate |
|---|---|---|---|---|---|
| Histoplasma | Septate hyphae | Small yeasts | 2-4 | Mississippi/Ohio valleys | 5-15% |
| Blastomyces | Septate hyphae | Broad-based budding | 8-15 | Great Lakes, Southeast | 10-40% |
| Coccidioides | Septate hyphae | Spherules | 20-200 | Southwest US, Mexico | 1-5% |
| Paracoccidioides | Septate hyphae | Multiple budding | 5-60 | South America | 5-27% |
| Sporothrix | Septate hyphae | Cigar-shaped yeasts | 2-6 | Worldwide | <1% |
The dimorphic switch correlates directly with virulence activation, where tissue forms express adhesins, proteases, and immune modulators absent in environmental forms, creating pathogen-specific therapeutic targets for next-generation antifungal development.
🌡️ Temperature-Driven Transformation: The Dimorphic Switch
🎯 Diagnostic Pattern Recognition: The Morphological Detective Work
📌 Remember: BUDDING-PSEUDO-TRUE - Budding yeasts (Candida), Pseudohyphae with constrictions (Candida), True septate hyphae (molds), distinguish by branching patterns and septation presence
Clinical morphological identification follows systematic algorithmic approaches that combine direct microscopy, culture characteristics, and biochemical testing to achieve >95% accuracy for common pathogenic fungi within 48-72 hours.
- Direct Microscopy Patterns
- KOH preparation: Dissolves host tissue, preserves fungal elements
- Calcofluor white: Binds chitin, fluorescent visualization
- Gram stain: Fungi appear gram-positive with thick walls
- Sensitivity: 40-60% for yeasts, 80-90% for molds
- Specificity: >95% when morphology is classic
- Turnaround time: 15-30 minutes
- Culture-Based Identification Framework
- Colony morphology: Color, texture, growth rate, reverse pigmentation
- Microscopic morphology: Conidial arrangement, septation patterns
- Biochemical tests: Germ tube, urease, carbohydrate assimilation
- Germ tube test: 95% sensitive for C. albicans
- Urease test: Positive in Cryptococcus (>90%)
- Chromogenic media: Species-specific color reactions
⭐ Clinical Pearl: Rapid diagnostic algorithms reduce time to appropriate therapy from 72-96 hours to 24-48 hours, decreasing mortality in invasive candidiasis by 15-25% and invasive aspergillosis by 20-30%
| Morphological Feature | Organism Group | Key Characteristics | Clinical Significance | Diagnostic Accuracy |
|---|---|---|---|---|
| Budding yeasts | Candida species | 4-6 μm, oval, pseudohyphae | Bloodstream infections | 85-95% |
| Encapsulated yeasts | Cryptococcus | 5-20 μm, thick capsule | CNS infections | >95% |
| Septate hyphae | Aspergillus | 45° branching, septate | Pulmonary infections | 80-90% |
| Nonseptate hyphae | Mucormycetes | 90° branching, ribbon-like | Rhinocerebral disease | 70-85% |
| Spherules | Coccidioides | 20-200 μm, endospores | Pulmonary/disseminated | >95% |
- MALDI-TOF MS: Protein fingerprinting, 90-95% accuracy
- Molecular sequencing: ITS region analysis, >99% accuracy
- Antigen detection: Galactomannan, β-D-glucan, cryptococcal antigen
- Galactomannan: 70% sensitive for invasive aspergillosis
- β-D-glucan: 80% sensitive for invasive candidiasis
- Cryptococcal antigen: >95% sensitive for cryptococcosis
💡 Master This: Morphological pattern recognition combined with clinical context (immunosuppression, geographic exposure, organ involvement) provides rapid diagnostic framework that enables targeted antifungal therapy before confirmatory testing completion
Pattern recognition mastery enables clinicians to distinguish >50 clinically relevant fungal species through systematic morphological analysis, transforming microscopic observations into life-saving therapeutic decisions within critical diagnostic windows.
🎯 Diagnostic Pattern Recognition: The Morphological Detective Work
⚔️ Pathogenesis Battleground: Host-Fungal Combat Strategies

📌 Remember: ADHESION-INVASION-EVASION - Fungi must Adhere to host surfaces, Invade tissue barriers, and Evade immune responses through specific molecular mechanisms at each stage
Successful fungal infection requires overcoming multiple host defense layers: physical barriers (skin, mucosa), innate immunity (neutrophils, macrophages), and adaptive immunity (T-cells, antibodies), with different fungi exploiting specific immune defects.
-
Virulence Factor Arsenal
- Adhesins: Enable binding to host cell receptors
- Invasins: Facilitate tissue penetration and dissemination
- Immune modulators: Suppress or misdirect host responses
- Complement inhibitors: Block classical pathway activation
- Cytokine mimics: Alter inflammatory responses
- Antigen masking: Hide from immune recognition
-
Host Defense Mechanisms
- Neutrophil responses: Phagocytosis, oxidative burst, NETs
- Macrophage activation: Antigen presentation, cytokine production
- T-cell immunity: Th1/Th17 responses crucial for fungal clearance
- Th1 cells: Produce IFN-γ, activate macrophages
- Th17 cells: Produce IL-17, recruit neutrophils
- Regulatory T-cells: Can impair antifungal responses
⭐ Clinical Pearl: Neutropenia (<500 cells/μL) increases invasive fungal infection risk by 50-100 fold, while corticosteroid therapy (>20mg prednisone daily for >3 weeks) impairs macrophage function and T-cell responses
| Risk Factor | Mechanism | Fungal Predisposition | Relative Risk | Mortality Impact |
|---|---|---|---|---|
| Neutropenia | Impaired phagocytosis | Candida, Aspergillus | 50-100x | 30-90% |
| Corticosteroids | T-cell suppression | Pneumocystis, Cryptococcus | 10-50x | 20-60% |
| HIV/AIDS | CD4+ T-cell depletion | Cryptococcus, Histoplasma | 100-1000x | 10-40% |
| Diabetes | Neutrophil dysfunction | Mucormycetes, Candida | 5-10x | 15-50% |
| Organ transplant | Multi-modal immunosuppression | All opportunistic fungi | 20-100x | 25-75% |
- Candida: Biofilm formation, phenotypic switching, tissue invasion
- Aspergillus: Angioinvasion, tissue necrosis, hematogenous spread
- Cryptococcus: Capsule-mediated immune evasion, CNS tropism
- Capsule thickness: Correlates with virulence
- Melanin production: Protects against oxidative stress
- Urease activity: Facilitates CNS invasion
💡 Master This: Immune status assessment guides antifungal prophylaxis decisions - neutropenia duration >10 days requires mold-active prophylaxis, while CD4+ counts <100 cells/μL necessitate Pneumocystis prophylaxis
Understanding host-pathogen interactions enables personalized antifungal strategies where immune status, exposure history, and species-specific virulence determine optimal prophylactic and therapeutic approaches for individual patient risk profiles.
⚔️ Pathogenesis Battleground: Host-Fungal Combat Strategies
🧪 Therapeutic Arsenal: Antifungal Combat Strategies

📌 Remember: POLYENE-AZOLE-ECHINO - Polyenes bind ergosterol (membrane), Azoles block ergosterol synthesis (14α-demethylase), Echinocandins inhibit β-glucan synthesis (cell wall)
Antifungal selection requires matching drug spectrum to likely pathogens, considering tissue penetration, drug interactions, and resistance patterns that vary significantly between fungal species and geographic regions.
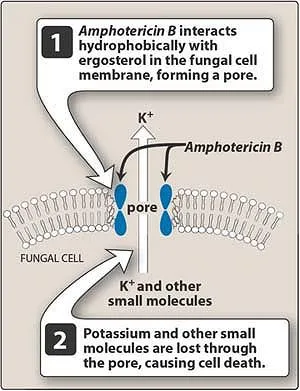
- Polyene Antifungals
- Mechanism: Bind ergosterol, create membrane pores
- Spectrum: Broad, including Candida, Aspergillus, Mucormycetes
- Resistance: Rare (<5%), involves ergosterol modifications
- Amphotericin B: Gold standard for invasive mycoses
- Liposomal formulations: Reduced nephrotoxicity
- Dosing: 3-5 mg/kg/day for invasive infections
- Azole Antifungals
- Mechanism: Inhibit 14α-demethylase (CYP51), block ergosterol synthesis
- Spectrum: Candida, Aspergillus, dermatophytes, endemic fungi
- Resistance: Increasing, especially fluconazole-resistant Candida
- Fluconazole: Limited mold activity, excellent CNS penetration
- Voriconazole: Broad spectrum, first-line for aspergillosis
- Isavuconazole: Fewer drug interactions, similar efficacy
⭐ Clinical Pearl: Echinocandins achieve >90% response rates in candidemia with minimal toxicity, making them first-line therapy for invasive candidiasis in non-neutropenic patients
| Drug Class | Mechanism | Spectrum | Resistance Rate | Major Toxicity | Cost Factor |
|---|---|---|---|---|---|
| Polyenes | Membrane binding | Broad | <5% | Nephrotoxicity | High |
| Azoles | Ergosterol synthesis | Broad | 5-30% | Hepatotoxicity | Moderate |
| Echinocandins | Cell wall synthesis | Candida/Aspergillus | <10% | Minimal | High |
| Flucytosine | DNA/RNA synthesis | Limited | 10-50% | Bone marrow | Low |
| Allylamines | Squalene epoxidase | Dermatophytes | <5% | Hepatotoxicity | Moderate |
- Synergistic combinations: Amphotericin B + flucytosine (cryptococcosis)
- Salvage therapy: Voriconazole + echinocandin (refractory aspergillosis)
- Sequential therapy: Echinocandin → azole (candidemia step-down)
- Combination benefits: Enhanced efficacy, reduced resistance
- Monitoring requirements: Drug levels, toxicity assessment
- Duration: Typically 14-42 days depending on infection
💡 Master This: Therapeutic drug monitoring for voriconazole (target trough 1-5.5 mg/L) and flucytosine (target 25-100 mg/L) optimizes efficacy while preventing concentration-dependent toxicity
Antifungal stewardship programs implementing species-directed therapy, therapeutic drug monitoring, and duration optimization reduce treatment costs by 30-50% while improving clinical outcomes and minimizing resistance development.
🧪 Therapeutic Arsenal: Antifungal Combat Strategies
🌐 Global Mycological Surveillance: Emerging Threats and Resistance
📌 Remember: ESCAPE-FUNGI - Emerging species, Surveillance networks, Cross-resistance patterns, Antifungal stewardship, Prevention strategies, Epidemiological tracking
Contemporary mycological threats include multidrug-resistant Candida auris, azole-resistant Aspergillus fumigatus, and emerging Candida species with intrinsic resistance patterns that challenge standard therapeutic approaches.
- Candida auris: The Superbug Threat
- Multidrug resistance: 90% fluconazole-resistant, 35% amphotericin-resistant
- Healthcare transmission: Persistent environmental contamination
- Mortality rates: 30-60% in invasive infections
- First identified: 2009 in Japan
- Global spread: >40 countries by 2023
- Outbreak potential: Healthcare facility clusters
- Azole-Resistant Aspergillus fumigatus
- Environmental resistance: Agricultural azole use drives selection
- Cross-resistance: Medical and agricultural azoles
- Geographic hotspots: Netherlands (20%), India (>50%)
- TR34/L98H mutation: Most common resistance mechanism
- Clinical impact: Treatment failure rates >50%
- Detection methods: Molecular screening required
⭐ Clinical Pearl: Candida auris requires contact precautions and enhanced environmental cleaning with chlorine-based disinfectants due to exceptional environmental persistence and healthcare transmission potential
| Emerging Threat | Resistance Pattern | Geographic Distribution | Mortality Rate | Detection Challenge |
|---|---|---|---|---|
| C. auris | MDR (90% azole-resistant) | Global, healthcare | 30-60% | Misidentification |
| A. fumigatus | Azole-resistant (5-50%) | Europe, Asia | 50-90% | Requires molecular testing |
| C. glabrata | Echinocandin-resistant (5-15%) | North America | 20-40% | Susceptibility testing |
| Mucormycetes | Intrinsic azole resistance | Worldwide | 40-80% | Rapid progression |
| Scedosporium | Multi-azole resistant | Global | 30-70% | Limited treatment options |
- ARTEMIS Global: Antifungal susceptibility monitoring
- SENTRY Program: Resistance trend tracking
- WHO Priority Pathogens: Critical resistance threats
- Data collection: >100 countries participating
- Resistance reporting: Annual trend analysis
- Clinical guidelines: Evidence-based updates

- Prevention and Control Strategies
- Antifungal stewardship: Optimize usage patterns
- Infection control: Prevent healthcare transmission
- Environmental monitoring: Agricultural and clinical settings
- Stewardship programs: 20-40% reduction in resistance
- Rapid diagnostics: Reduce inappropriate therapy
- Combination therapy: Prevent resistance development
💡 Master This: Rapid molecular diagnostics identifying resistance genes (TR34/L98H in Aspergillus, ERG11 mutations in Candida) enable immediate therapeutic optimization before phenotypic susceptibility results become available
Global mycological surveillance networks provide real-time resistance intelligence that guides empirical therapy selection, infection control measures, and antifungal development priorities essential for maintaining therapeutic effectiveness against evolving fungal threats.
🌐 Global Mycological Surveillance: Emerging Threats and Resistance
🎯 Clinical Mastery Toolkit: Rapid Mycological Decision Framework
📌 Remember: RAPID-FUNGI - Risk assessment, Anatomic site, Patient factors, Immunosuppression, Diagnostic approach, Fungal likelihood, Urgent therapy, Narrow spectrum, Guided therapy, Infection control
- Essential Clinical Arsenal
- High-risk populations: Neutropenia, transplant, ICU patients
- Diagnostic priorities: Blood cultures, imaging, biomarkers
- Empirical therapy: Species-directed, site-specific selection
- Candidemia: Echinocandin first-line (>90% efficacy)
- Invasive aspergillosis: Voriconazole (70-80% response)
- Mucormycosis: Amphotericin B (40-60% survival)
| Clinical Scenario | First-Line Therapy | Alternative Options | Duration | Monitoring |
|---|---|---|---|---|
| Candidemia | Caspofungin 70mg→50mg | Fluconazole (if susceptible) | 14 days post-clearance | Blood cultures q48h |
| Invasive Aspergillosis | Voriconazole 6mg/kg q12h | Isavuconazole, AmB | 6-12 weeks | Drug levels, imaging |
| Cryptococcal Meningitis | AmB + Flucytosine | Fluconazole high-dose | 10 weeks total | CSF pressure, cultures |
| Mucormycosis | Liposomal AmB 5mg/kg | Posaconazole, surgery | Until resolution | Imaging, debridement |
| PCP Pneumonia | TMP-SMX high-dose | Pentamidine, atovaquone | 21 days | Oxygenation, steroids |
💡 Master This: Biomarker-guided therapy using β-D-glucan (>80 pg/mL suggests invasive fungal infection) and galactomannan (>0.5 indicates aspergillosis) enables earlier diagnosis and targeted treatment before culture confirmation
Clinical mycological expertise transforms complex diagnostic challenges into systematic therapeutic successes through evidence-based decision frameworks that optimize patient outcomes while minimizing resistance development and healthcare costs.
🎯 Clinical Mastery Toolkit: Rapid Mycological Decision Framework
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more
Have doubts about this lesson?
Ask Rezzy, your AI Study Mate, to explain anything you didn't understand
Everything you need for NEET-PG prep
Get full Oncourse access with lessons, practice questions, flashcards and AI study tools.
Scan to download app






















