Nephrology

On this page
🔬 Glomerular Architecture: The Kidney's Precision Filtration Network
The glomerulus is medicine's most elegant molecular sieve, filtering 180 liters daily while retaining essential proteins through nanoscale precision-yet when this barrier fails, the resulting syndromes reveal themselves through distinctive urinary fingerprints. You'll master how glomerular architecture dictates filtration selectivity, decode nephritic versus nephrotic patterns from first principles, navigate the diagnostic matrix linking clinical presentation to specific diseases, and deploy targeted immunosuppressive and supportive therapies while recognizing when glomerular injury signals systemic disease requiring broader intervention.

The glomerular filtration barrier operates as a three-layer security system with distinct molecular size and charge selectivity:
-
Fenestrated Endothelium (Layer 1)
- Pore diameter: 70-100 nm
- Blocks cellular elements and large proteins
- Maintains negative surface charge via glycocalyx
- Prevents albumin adhesion through charge repulsion
- Fenestrations occupy 20-50% of surface area
- Missing diaphragms unlike other capillaries
-
Glomerular Basement Membrane (Layer 2)
- Thickness: 300-350 nm in adults
- Type IV collagen provides structural framework
- Laminin and proteoglycans create charge barrier
- Heparan sulfate contributes negative charge density
- Molecular weight cutoff: 40-60 kDa
- Thickens with age and diabetes (↑500-800 nm)
-
Podocyte Foot Processes (Layer 3)
- Slit diaphragm width: 40 nm precisely
- Nephrin and podocin proteins form filtration slits
- Actin cytoskeleton maintains structural integrity
- Foot process effacement indicates podocyte injury
- 2,500+ slit diaphragms per podocyte
- Covers 60% of GBM surface area
📌 Remember: GBM-FEN - Glomerular Basement Membrane, Fenestrated Endothelium, Nephrin slits. Three barriers working together: size exclusion (>60 kDa blocked), charge repulsion (negative proteins repelled), and slit filtration (40 nm maximum).

| Filtration Component | Primary Function | Pore Size | Charge | Disease Association | Clinical Marker |
|---|---|---|---|---|---|
| Fenestrated Endothelium | Cellular exclusion | 70-100 nm | Negative | Endotheliosis | Microhematuria |
| Glomerular Basement Membrane | Size/charge barrier | 40-60 kDa | Negative | Alport syndrome | Proteinuria |
| Podocyte Slit Diaphragm | Final filtration | 40 nm | Neutral | Minimal change | Nephrotic syndrome |
| Mesangial Matrix | Structural support | Variable | Negative | IgA nephropathy | Mesangial expansion |
| Bowman's Capsule | Urine collection | N/A | Neutral | Crescentic GN | Crescent formation |
The glomerular capillary network demonstrates remarkable hemodynamic precision through specialized vascular architecture:
-
Afferent Arteriole Regulation
- Diameter: 15-20 μm at glomerular entrance
- Autoregulation maintains constant flow between 80-180 mmHg
- Myogenic response prevents pressure transmission
- Tubuloglomerular feedback via macula densa
- Angiotensin II causes preferential constriction
- NSAID inhibition reduces GFR by 15-25%
-
Glomerular Capillary Pressure
- Hydrostatic pressure: 45-60 mmHg (highest in body)
- Oncotic pressure: 25-35 mmHg opposing filtration
- Net filtration pressure: 10-25 mmHg driving force
- Pressure drops 5-10 mmHg along capillary length
- Filtration fraction: 20% of renal plasma flow
- Single nephron GFR: 60-80 nL/min
💡 Master This: Glomerular pressure regulation determines filtration efficiency. Afferent dilation or efferent constriction increases GFR, while afferent constriction or efferent dilation decreases GFR. ACE inhibitors preferentially dilate efferent arterioles, reducing intraglomerular pressure by 10-15 mmHg while maintaining systemic blood pressure.
Connect this architectural foundation through glomerular pathophysiology to understand how structural damage creates specific clinical syndromes.
🔬 Glomerular Architecture: The Kidney's Precision Filtration Network
⚙️ Filtration Dynamics: The Molecular Sorting Engine
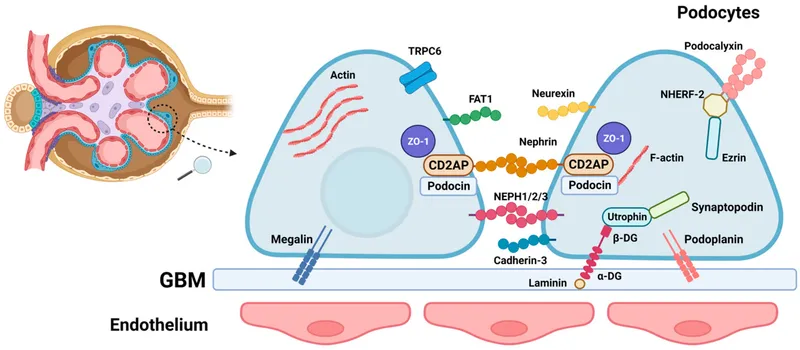
The Starling forces governing glomerular filtration create a unique high-pressure system optimized for continuous plasma processing:
-
Hydrostatic Pressure Gradient
- Glomerular capillary pressure: 50 ± 5 mmHg
- Bowman's space pressure: 10 ± 2 mmHg
- Net hydrostatic driving force: 40 mmHg
- 3x higher than systemic capillaries
- Maintained by efferent arteriole resistance
- Autoregulation preserves pressure despite ±30% systemic BP changes
-
Oncotic Pressure Opposition
- Afferent oncotic pressure: 25 mmHg (normal protein)
- Efferent oncotic pressure: 35 mmHg (concentrated)
- Average opposing force: 30 mmHg
- Increases along capillary as water filters out
- Hypoalbuminemia reduces opposition by 40-60%
- Hyperfiltration occurs when oncotic pressure <20 mmHg
📌 Remember: HOPE for filtration - Hydrostatic pressure Outward, Oncotic pressure Prevents, Equilibrium determines GFR. Net filtration pressure = (Hydrostatic in - Hydrostatic out) - (Oncotic in - Oncotic out) = (50-10) - (25-0) = 15 mmHg average driving force.

Molecular selectivity depends on size, charge, and shape characteristics that determine filtration coefficients:
| Molecule | Molecular Weight | Radius (nm) | Charge | Filtration Fraction | Clinical Significance |
|---|---|---|---|---|---|
| Water | 18 Da | 0.1 | Neutral | 1.0 | Free filtration |
| Glucose | 180 Da | 0.36 | Neutral | 1.0 | Diabetic spillover >180 mg/dL |
| Inulin | 5,200 Da | 1.48 | Neutral | 1.0 | GFR measurement standard |
| Myoglobin | 17,800 Da | 1.9 | Positive | 0.75 | Rhabdomyolysis marker |
| Albumin | 67,000 Da | 3.6 | Negative | 0.0001 | Proteinuria indicator |
| IgG | 150,000 Da | 5.5 | Variable | <0.0001 | Glomerular damage marker |
- Molecules <1.8 nm radius filter freely (100%)
- Progressive restriction from 1.8-4.0 nm (50-1%)
- Virtual exclusion >4.0 nm radius (<0.1%)
- Albumin radius 3.6 nm explains minimal normal filtration
- Hemoglobin 3.2 nm appears with intravascular hemolysis
- Transferrin 4.0 nm indicates severe barrier damage
- Charge-Selective Mechanisms
- Negative charges repelled by heparan sulfate in GBM
- Albumin filtration ↑1000-fold when charge neutralized
- Cationic proteins filter 5-10x more readily than anionic
- Explains why lysozyme (positive) appears early in disease
- Diabetic nephropathy loses charge selectivity first
- Minimal change disease preserves size but loses charge barrier
⭐ Clinical Pearl: Proteinuria patterns reveal filtration barrier damage location. Selective proteinuria (albumin predominant) suggests charge barrier loss with preserved size selectivity (minimal change disease). Non-selective proteinuria (mixed proteins) indicates structural GBM damage (focal segmental glomerulosclerosis). Microalbuminuria 30-300 mg/day detects early diabetic nephropathy with 85% sensitivity.
💡 Master This: Filtration fraction = GFR/Renal Plasma Flow normally equals 20%. Increased filtration fraction (>25%) suggests efferent arteriole constriction (angiotensin II, NSAIDs), while decreased fraction (<15%) indicates afferent constriction or reduced glomerular pressure. This ratio guides therapeutic targeting of specific arteriolar segments.
Connect these filtration principles through pattern recognition frameworks to understand how different diseases create characteristic urinalysis findings.
⚙️ Filtration Dynamics: The Molecular Sorting Engine
🎯 Pattern Recognition: The Glomerular Disease Decoder
Proteinuria Pattern Recognition provides the primary diagnostic framework:
-
Nephrotic-Range Proteinuria (>3.5 g/day)
- See: Foamy urine, periorbital edema, hypoalbuminemia <3.0 g/dL
- Think: Podocyte disease with charge barrier loss
- Top Differentials: Minimal change (children 85%), FSGS (adults 40%), membranous (adults 30%)
- Selective proteinuria suggests minimal change disease
- Non-selective proteinuria indicates structural damage
- Albumin:creatinine ratio >2200 mg/g confirms nephrotic range
-
Sub-Nephrotic Proteinuria (150 mg - 3.5 g/day)
- See: Mild edema, preserved albumin >3.5 g/dL
- Think: Early glomerular damage or tubular disease
- Top Differentials: Early diabetic nephropathy, hypertensive nephrosclerosis, chronic GN
- Microalbuminuria 30-300 mg/day suggests diabetic nephropathy
- ACR 30-300 mg/g indicates cardiovascular risk
- Progressive increase over months suggests glomerular disease
📌 Remember: FOAM for nephrotic syndrome - Foamy urine, Oedema (periorbital), Albumin low (<3.0 g/dL), Massive proteinuria (>3.5 g/day). Plus hyperlipidemia (cholesterol >250 mg/dL) and hypercoagulability (increased thrombosis risk 6-fold).
Hematuria Pattern Analysis reveals glomerular versus non-glomerular bleeding:
| Hematuria Type | RBC Morphology | Proteinuria | Casts | Clinical Context | Diagnostic Yield |
|---|---|---|---|---|---|
| Glomerular | Dysmorphic >80% | Usually present | RBC casts | Systemic symptoms | 95% specific |
| Non-glomerular | Isomorphic >80% | Minimal | Rare | Urological symptoms | 90% specific |
| Mixed | 50-80% dysmorphic | Variable | Occasional | Complex presentation | Requires imaging |
| Pseudo-hematuria | No RBCs | Absent | None | Medications/foods | 100% specific |
- Dysmorphic RBCs >80% with acanthocytes pathognomonic
- RBC casts present in 60-80% of acute glomerulonephritis
- Proteinuria accompanies hematuria in >90% of cases
- Cola-colored urine suggests acute post-infectious GN
- Episodic gross hematuria with URI suggests IgA nephropathy
- Persistent microscopic hematuria requires glomerular evaluation
- Acute Nephritic Syndrome Recognition
- See: Gross hematuria, hypertension, oliguria, edema
- Think: Inflammatory glomerular disease with reduced GFR
- Laboratory: ↑Creatinine, ↓C3, ↑ASO (post-infectious)
- GFR reduction >50% over days to weeks
- Sodium retention causes rapid weight gain (>2 kg/week)
- Hypertension in 80-90% due to volume expansion

Rapidly Progressive Glomerulonephritis (RPGN) requires immediate recognition:
-
Clinical Presentation
- Creatinine doubling within 3 months or 50% GFR loss
- Oliguria (<400 mL/day) in 70% of cases
- Systemic symptoms: fever, weight loss, arthritis
- Pulmonary-renal syndrome suggests ANCA vasculitis
- Hemoptysis + hematuria = Goodpasture's until proven otherwise
- Rash + arthritis + nephritis = lupus nephritis consideration
-
Laboratory Patterns
- Crescents on biopsy in >50% of glomeruli
- ANCA positive in 60% (c-ANCA = GPA, p-ANCA = MPA)
- Anti-GBM antibodies in 5-10% (Goodpasture's)
- Double-positive ANCA + anti-GBM carries worst prognosis
- ANA positive suggests lupus nephritis
- Low C3/C4 indicates complement consumption
⭐ Clinical Pearl: RPGN triad - rapid GFR decline (>50% in 3 months), active urinary sediment (RBC casts), and systemic inflammation (ESR >50). Requires urgent nephrology consultation and kidney biopsy within 24-48 hours. Delayed diagnosis reduces renal recovery from 70% to <20%.
💡 Master This: Urine sediment provides the roadmap to glomerular disease. RBC casts = active glomerulonephritis requiring immediate evaluation. WBC casts = tubulointerstitial disease or severe glomerular inflammation. Fatty casts = nephrotic syndrome with lipiduria. Broad waxy casts = chronic kidney disease with nephron loss.
Connect these recognition patterns through systematic comparison frameworks to distinguish between similar glomerular presentations.
🎯 Pattern Recognition: The Glomerular Disease Decoder
🔬 Differential Diagnosis: The Glomerular Disease Matrix
Primary Glomerular Disease Discrimination focuses on presentation patterns and demographics:
| Disease | Age Peak | Proteinuria | Hematuria | Hypertension | Progression | Key Discriminator |
|---|---|---|---|---|---|---|
| Minimal Change | 2-6 years | Massive (>3.5g) | Rare | Uncommon | Excellent | Steroid responsive 95% |
| FSGS | 20-40 years | Variable | Common | Frequent | Poor | African American 70% |
| Membranous | 40-60 years | Massive | Rare | Mild | Variable | Anti-PLA2R positive 80% |
| IgA Nephropathy | 20-30 years | Mild-moderate | Episodic gross | Common | Slow | Synpharyngitic hematuria |
| Post-infectious GN | 5-15 years | Moderate | Gross | Severe | Good | Low C3, high ASO |
- Demographics: 85% of childhood nephrotic syndrome
- Presentation: Sudden onset massive proteinuria, normal GFR
- Laboratory: Selective proteinuria, normal complement
- Steroid response in 95% within 8 weeks
- Relapse rate 50-70% but maintains steroid sensitivity
- Normal kidney biopsy on light microscopy
- Focal Segmental Glomerulosclerosis (FSGS)
- Demographics: 40% of adult nephrotic syndrome, African American predominance
- Presentation: Hypertension in 80%, reduced GFR at diagnosis
- Laboratory: Non-selective proteinuria, steroid resistance >80%
- ESRD progression in 50% within 10 years
- Recurrence in 30% of transplants
- Genetic forms account for 10-15% of cases
📌 Remember: FSGS-MCD differentiation - Frequent hypertension, Steroid resistance, GFR reduced, Sclerosis on biopsy versus Massive proteinuria only, Childhood onset, Dramatic steroid response. Age >40 with nephrotic syndrome = membranous nephropathy until proven otherwise.
Secondary Glomerular Disease Recognition requires identifying underlying systemic conditions:
-
Diabetic Nephropathy Progression
- Stage 1: Hyperfiltration (GFR >150 mL/min)
- Stage 2: Microalbuminuria (30-300 mg/day)
- Stage 3: Macroalbuminuria (>300 mg/day)
- Kimmelstiel-Wilson nodules pathognomonic on biopsy
- Retinopathy present in >90% with nephropathy
- ACE inhibitor reduces progression by 40-50%
-
Lupus Nephritis Classification
- Class I-II: Minimal/mesangial (normal GFR, mild proteinuria)
- Class III-IV: Focal/diffuse endocapillary (active nephritis)
- Class V: Membranous (nephrotic syndrome)
- Class IV has worst prognosis (30% ESRD at 10 years)
- Activity index guides immunosuppression intensity
- Chronicity index predicts irreversible damage

ANCA-Associated Vasculitis differentiation impacts treatment selection:
-
Granulomatosis with Polyangiitis (GPA)
- c-ANCA/PR3 positive in 95% of generalized disease
- Upper respiratory tract involvement (sinusitis, saddle nose)
- Pulmonary nodules with cavitation characteristic
- Renal involvement in 80% of generalized cases
- Cyclophosphamide induction followed by maintenance therapy
- Relapse rate 50% within 5 years
-
Microscopic Polyangiitis (MPA)
- p-ANCA/MPO positive in 70% of cases
- Pulmonary-renal syndrome without upper respiratory involvement
- Rapidly progressive course with poor prognosis if untreated
- Alveolar hemorrhage in 30% of cases
- Plasma exchange for severe pulmonary involvement
- Maintenance remission requires long-term immunosuppression
⭐ Clinical Pearl: Anti-GBM disease (Goodpasture's) presents with linear IgG on immunofluorescence, anti-GBM antibodies >20 EU/mL, and pulmonary-renal syndrome in 60%. Plasma exchange must begin within 24-48 hours for renal recovery. Oliguria at presentation predicts irreversible renal failure in >90% of cases.
💡 Master This: Complement levels guide differential diagnosis. Low C3 suggests post-infectious GN, MPGN, or lupus nephritis. Low C4 with normal C3 suggests cryoglobulinemia. Normal complement with RPGN points toward ANCA vasculitis or anti-GBM disease. Alternative pathway activation (low C3, normal C4) occurs in C3 glomerulopathy.
Connect these discrimination principles through evidence-based treatment algorithms to optimize therapeutic outcomes.
🔬 Differential Diagnosis: The Glomerular Disease Matrix
⚕️ Treatment Algorithms: The Therapeutic Precision Matrix
Nephrotic Syndrome Treatment Protocols follow evidence-based response patterns:
-
Minimal Change Disease Management
- Initial Treatment: Prednisone 1 mg/kg/day (max 80 mg) for 4-6 weeks
- Response Rate: 95% achieve remission within 8 weeks
- Taper Protocol: Reduce by 10 mg weekly once remission achieved
- Relapse rate: 50-70% within 2 years
- Frequent relapsers: >2 relapses in 6 months
- Steroid-dependent: Relapse during taper or within 2 weeks of stopping
-
FSGS Treatment Strategy
- Primary FSGS: Steroid trial 1 mg/kg/day for 16 weeks
- Response Rate: 20-30% complete remission, 40% partial
- Second-line: Calcineurin inhibitors (cyclosporine 3-5 mg/kg/day)
- Rituximab shows 40% response in steroid-resistant cases
- Mycophenolate alternative with fewer side effects
- ACE inhibitors mandatory for proteinuria reduction
| Treatment | Indication | Dosing | Response Rate | Time to Response | Major Side Effects |
|---|---|---|---|---|---|
| Prednisone | MCD, FSGS | 1 mg/kg/day | 95% MCD, 30% FSGS | 4-8 weeks | Infection, osteoporosis |
| Cyclosporine | Steroid-resistant | 3-5 mg/kg/day | 60-70% | 3-6 months | Nephrotoxicity, hypertension |
| Rituximab | Relapsing MCD | 375 mg/m² x4 | 80% | 2-4 months | Infection, hypogammaglobulinemia |
| Mycophenolate | FSGS maintenance | 1-2 g/day | 40-50% | 6-12 months | GI upset, leukopenia |
| Cyclophosphamide | Severe FSGS | 2 mg/kg/day | 50-60% | 3-6 months | Infertility, malignancy |
RPGN Emergency Management requires immediate intervention to preserve renal function:
-
Induction Therapy Protocol
- Cyclophosphamide: 2 mg/kg/day oral or 15 mg/kg IV monthly
- Methylprednisolone: 1 g daily x 3 days, then prednisone 1 mg/kg
- Plasma exchange: 7-14 sessions for severe disease or anti-GBM
- Response rate: 70-80% if creatinine <5.7 mg/dL
- Dialysis independence: 60% if treated within 2 weeks
- Maintenance therapy: Azathioprine or methotrexate
-
ANCA Vasculitis Specific Protocols
- Severe disease: Cyclophosphamide + steroids for 3-6 months
- Limited disease: Methotrexate + steroids alternative
- Maintenance: Azathioprine 2 mg/kg/day or methotrexate 25 mg/week
- Rituximab non-inferior to cyclophosphamide for induction
- Remission rate: 85-90% with standard protocols
- Relapse prevention: Maintenance therapy for 18-24 months
Lupus Nephritis Treatment stratified by histological class and activity:
-
Class III/IV Lupus Nephritis
- Induction: Mycophenolate 3 g/day or cyclophosphamide monthly
- Response Rate: 80% complete or partial remission at 6 months
- Maintenance: Mycophenolate 2 g/day for minimum 3 years
- Hydroxychloroquine mandatory unless contraindicated
- ACE inhibitors for proteinuria >0.5 g/day
- Renal flare rate: 15-20% despite maintenance therapy
-
Monitoring Parameters
- Proteinuria reduction >50% by 6 months predicts long-term response
- Complete remission: Proteinuria <0.5 g/day, normal GFR
- Partial remission: 50% proteinuria reduction, stable GFR
- Anti-dsDNA and C3/C4 levels guide treatment intensity
- Repeat biopsy if no response at 6 months
- Pregnancy planning requires stable remission for 6 months
⭐ Clinical Pearl: Treatment response in glomerular disease follows predictable timelines. Steroids show response in 4-8 weeks, calcineurin inhibitors in 3-6 months, cytotoxic agents in 3-6 months. No improvement by expected timeframe indicates treatment failure requiring protocol change or biopsy reassessment.
💡 Master This: Proteinuria reduction serves as the primary endpoint for treatment success. >50% reduction by 6 months predicts long-term renal survival. Complete remission (proteinuria <0.3 g/day) provides best long-term outcomes with 90% 10-year renal survival. Partial remission still offers significant protection compared to no response.
Connect these treatment principles through advanced integration concepts to understand multi-system disease management.
⚕️ Treatment Algorithms: The Therapeutic Precision Matrix
🌐 Multi-System Integration: The Glomerular-Systemic Interface
Cardiovascular-Renal Integration demonstrates how glomerular disease accelerates cardiovascular risk through multiple mechanisms:
-
Proteinuria-Cardiovascular Risk Relationship
- Albuminuria >30 mg/g increases CV mortality by 50%
- Nephrotic-range proteinuria carries 6-fold increased thrombotic risk
- GFR decline correlates with exponential CV risk increase
- eGFR 45-59: 20% increased CV mortality
- eGFR 30-44: 80% increased CV mortality
- eGFR <30: 300% increased CV mortality
-
Mechanistic Pathways
- Endothelial dysfunction from uremic toxins and inflammation
- Accelerated atherosclerosis via oxidative stress and dyslipidemia
- Left ventricular hypertrophy from volume overload and hypertension
- Mineral bone disorder contributes to vascular calcification
- Chronic inflammation (↑CRP, ↑IL-6) promotes plaque instability
- Hypercoagulable state increases thrombotic events by 400%
📌 Remember: CHAMP for CV risk in glomerular disease - Chronic inflammation, Hypertension, Albuminuria, Mineral bone disorder, Platelet dysfunction. Each 10 mL/min GFR decline increases CV mortality by 10-15%. Statin therapy reduces CV events by 25% regardless of cholesterol levels.
Hematologic Complications arise from bone marrow suppression, uremic bleeding, and hypercoagulability:
| Hematologic Issue | Mechanism | Prevalence | Clinical Impact | Management | Monitoring |
|---|---|---|---|---|---|
| Anemia | EPO deficiency | 90% if GFR <30 | Fatigue, LVH | ESA, iron | Hgb target 10-11 |
| Bleeding | Platelet dysfunction | 50% uremic patients | Surgical risk | Desmopressin | Bleeding time |
| Thrombosis | Nephrotic syndrome | 25% nephrotic | PE, DVT, RVT | Anticoagulation | D-dimer, imaging |
| Bone Disease | Mineral disorder | 80% CKD 3-5 | Fractures, pain | Phosphate binders | PTH, Ca, PO4 |
- Platelet adhesion impaired by uremic toxins
- von Willebrand factor dysfunction reduces hemostasis
- Bleeding time prolonged despite normal platelet count
- Desmopressin 0.3 μg/kg improves bleeding time for 4-8 hours
- Cryoprecipitate provides temporary hemostasis for surgery
- Dialysis corrects bleeding tendency within 24-48 hours
- Nephrotic Hypercoagulability
- Antithrombin III loss in urine reduces anticoagulation
- Increased fibrinogen and factor VIII promote clotting
- Platelet aggregation enhanced by hyperlipidemia
- Renal vein thrombosis occurs in 5-15% of membranous nephropathy
- Pulmonary embolism risk increased 8-fold in nephrotic syndrome
- Prophylactic anticoagulation considered if albumin <2.0 g/dL
Immunologic System Interactions reveal how glomerular inflammation affects systemic immunity:
-
Complement System Dysregulation
- Classical pathway activation in lupus nephritis and post-infectious GN
- Alternative pathway dysregulation in C3 glomerulopathy and aHUS
- Lectin pathway involvement in IgA nephropathy progression
- C3 nephritic factor causes persistent C3 consumption
- Factor H mutations predispose to atypical HUS
- Eculizumab blocks terminal complement in aHUS with 90% response
-
Autoimmune Disease Associations
- ANCA vasculitis affects kidneys in 80% of generalized disease
- Lupus nephritis develops in 60% of SLE patients within 10 years
- Anti-GBM disease has pulmonary involvement in 60% of cases
- Molecular mimicry between infections and glomerular antigens
- HLA associations (DR15 in anti-GBM, DR3 in membranous)
- Environmental triggers (hydrocarbons, infections) precipitate disease
⭐ Clinical Pearl: Glomerular disease creates accelerated aging phenotype with premature cardiovascular disease, bone loss, immune dysfunction, and cognitive impairment. Comprehensive care addressing all complications improves survival more than isolated kidney treatment. Multidisciplinary approach reduces hospitalization by 30% and mortality by 20%.
💡 Master This: Proteinuria serves as both kidney disease marker and independent cardiovascular risk factor. ACE inhibitors provide dual benefit - renal protection through reduced proteinuria and cardiovascular protection through improved endothelial function. Target proteinuria <1 g/day for optimal outcomes in both systems.
Connect these integration principles through rapid mastery frameworks to develop comprehensive clinical expertise.
🌐 Multi-System Integration: The Glomerular-Systemic Interface
🎯 Clinical Mastery Arsenal: The Glomerular Disease Command Center
Essential Numbers Arsenal - memorize these critical thresholds for instant clinical application:
-
Proteinuria Thresholds
- Normal: <150 mg/day or <30 mg/g creatinine
- Microalbuminuria: 30-300 mg/day (diabetic nephropathy screening)
- Nephrotic range: >3.5 g/day or >2200 mg/g creatinine
- Selective proteinuria: Albumin selectivity index <0.1
- Massive proteinuria: >10 g/day suggests minimal change or membranous
- Proteinuria reduction >50% by 6 months = treatment success
-
GFR Critical Values
- Normal: >90 mL/min/1.73m² with normal kidney damage markers
- CKD Stage 3: 30-59 mL/min/1.73m² (nephrology referral)
- CKD Stage 4: 15-29 mL/min/1.73m² (RRT preparation)
- RPGN definition: 50% GFR loss in <3 months
- Dialysis urgency: GFR <10 or uremic symptoms
- Transplant evaluation: GFR <20 in appropriate candidates
📌 Remember: URGENT glomerular emergencies - Uremia (GFR <10), RPGN (50% GFR loss <3 months), Goodpasture's (anti-GBM +), Eclampsia (pregnancy), Nephrotic crisis (infection), Thrombosis (renal vein). Each requires <24 hour intervention for optimal outcomes.
Rapid Diagnostic Framework - systematic approach for efficient evaluation:
| Clinical Presentation | First-Line Tests | Key Discriminators | Urgent Actions | Specialist Referral |
|---|---|---|---|---|
| Nephrotic Syndrome | Urine protein, albumin, lipids | Age, steroid response | Infection screen | Pediatric: immediate |
| Acute Nephritis | CBC, BUN/Cr, C3/C4, ASO | Complement levels | BP control | <48 hours |
| RPGN | ANCA, anti-GBM, ANA | Antibody pattern | Urgent biopsy | <24 hours |
| Asymptomatic Hematuria | Urine microscopy, imaging | RBC morphology | Rule out malignancy | <2 weeks |
| Proteinuria + HTN | ACR, eGFR, diabetic screen | Diabetes history | ACE inhibitor | <1 month |
-
Steroid-Responsive Disease
- Week 2: 50% proteinuria reduction suggests responsiveness
- Week 4: Complete remission expected in minimal change disease
- Week 8: 95% of MCD patients achieve remission
- No response by week 8 = steroid resistance
- Relapse during taper = steroid dependence
- >2 relapses/6 months = frequent relapser
-
Immunosuppressive Therapy
- Month 3: Partial response (>25% proteinuria reduction)
- Month 6: Treatment success (>50% proteinuria reduction)
- Month 12: Complete remission (proteinuria <0.3 g/day)
- Cyclophosphamide: Maximum 6 months to avoid toxicity
- Rituximab: Response may delay 3-6 months
- CNI therapy: Monitor levels and renal function monthly
⭐ Clinical Pearl: Biopsy timing determines diagnostic yield and treatment success. Acute nephritis: biopsy within 48 hours if RPGN suspected. Nephrotic syndrome: biopsy if steroid resistant or atypical presentation. Chronic disease: biopsy guides prognosis and treatment intensity. Contraindications: bleeding risk, small kidneys (<9 cm), uncontrolled hypertension.
Complication Prevention Checklist - systematic approach to reduce morbidity:
-
Cardiovascular Protection
- ACE inhibitor/ARB for proteinuria >0.5 g/day
- Statin therapy for LDL >100 mg/dL or nephrotic syndrome
- Blood pressure target <130/80 in CKD patients
- Aspirin for primary prevention if bleeding risk low
- Smoking cessation reduces CV risk by 50%
- Exercise and dietary sodium <2 g/day
-
Infection Prevention
- Pneumococcal vaccine for nephrotic syndrome and immunosuppression
- Annual influenza vaccination for all CKD patients
- Live vaccines contraindicated during immunosuppressive therapy
- Prophylactic antibiotics for severe neutropenia (<500)
- PCP prophylaxis for high-dose steroids >3 months
- Monitor for opportunistic infections during treatment
💡 Master This: Glomerular disease outcomes depend on early recognition, appropriate treatment, and comprehensive management. Proteinuria reduction remains the primary target with >50% reduction providing significant renal protection. Multidisciplinary care addressing cardiovascular risk, bone disease, anemia, and complications improves long-term survival and quality of life.
🎯 Clinical Mastery Arsenal: The Glomerular Disease Command Center
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more
Have doubts about this lesson?
Ask Rezzy, your AI Study Mate, to explain anything you didn't understand
Everything you need for NEET-PG prep
Get full Oncourse access with lessons, practice questions, flashcards and AI study tools.
Scan to download app
























