Back
What Is a Zymogen? Definition, Function, and Examples in 2026
Learn about zymogens (enzyme precursors), their activation mechanisms, clinical significance, and key examples including digestive enzymes and blood coagulation factors for medical exams.

What Is a Zymogen? Definition, Function, and Examples in 2026
Zymogens are inactive enzyme precursors that play a crucial role in biochemical processes throughout the human body. Understanding zymogens is essential for medical students preparing for exams like NEET-PG, USMLE, and UKMLA, as they frequently appear in questions related to biochemistry, physiology, and pathology. This comprehensive guide explores everything you need to know about zymogens, their functions, and clinical significance.
What Is a Zymogen?
A zymogen (also called a proenzyme) is an inactive precursor form of an enzyme that requires specific activation to become catalytically active. The term "zymogen" derives from the Greek words "zyme" (meaning ferment or enzyme) and "gen" (meaning producing or creating).
Zymogens are synthesized as larger, inactive proteins that contain the complete enzyme structure plus additional peptide sequences. These additional sequences prevent the enzyme from being active until specific conditions are met, providing precise control over when and where enzymatic activity occurs.
Key Characteristics of Zymogens
Inactive state: Zymogens lack catalytic activity in their precursor form
Larger molecular weight: They are typically larger than their active enzyme counterparts
Specific activation mechanisms: Require proteolytic cleavage or other modifications to become active
Regulatory function: Allow cells to control enzyme activity spatially and temporally
Safety mechanism: Prevent premature enzyme activation that could damage tissues
Why Do Zymogens Exist?
The existence of zymogens serves several critical biological functions:
1. Temporal Control
Zymogens allow organisms to produce enzymes in advance but activate them only when needed. This is particularly important for digestive enzymes that could damage the cells producing them if activated prematurely.
2. Spatial Control
By requiring specific activation signals, zymogens ensure that enzymes become active only in appropriate locations. For example, digestive zymogens are activated only after leaving the pancreas.
3. Metabolic Efficiency
Producing inactive precursors allows for rapid enzyme activation when needed, without requiring new protein synthesis during critical processes like blood clotting.
4. Protective Mechanism
Zymogens protect tissues from potentially harmful enzymes. Pancreatic digestive enzymes, if activated within the pancreas, would cause severe tissue damage (pancreatitis).
Zymogen Activation Mechanisms
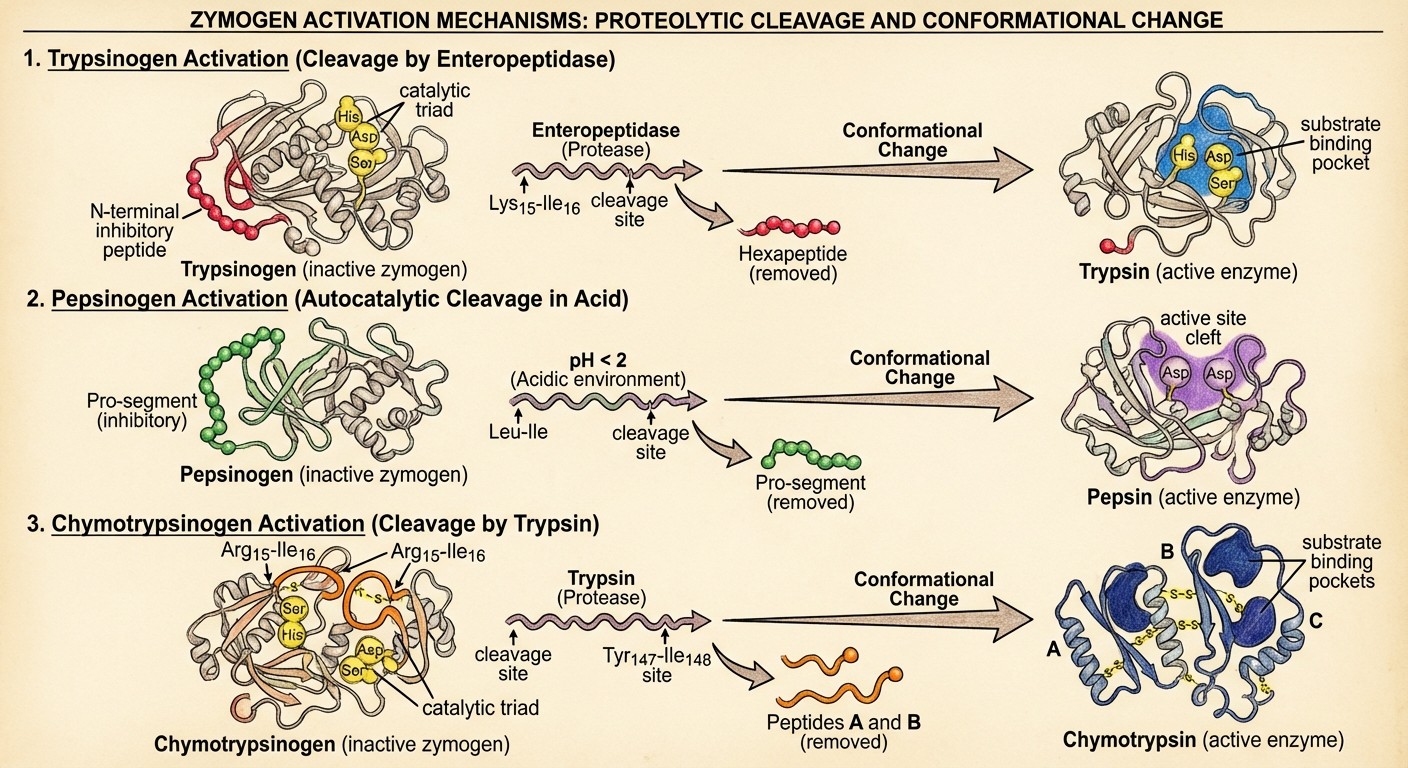
Zymogens undergo activation through several distinct mechanisms:
Proteolytic Cleavage
The most common activation mechanism involves the cleavage of specific peptide bonds by proteolytic enzymes. This process typically removes an inhibitory peptide sequence, exposing the active site of the enzyme.
Examples:
Pepsinogen → Pepsin (by acid and autocatalysis)
Trypsinogen → Trypsin (by enterokinase)
Chymotrypsinogen → Chymotrypsin (by trypsin)
Autocatalysis
Some zymogens can activate themselves once initial activation begins. This creates a positive feedback loop that rapidly amplifies enzyme activity.
Example: Once a small amount of pepsin is formed from pepsinogen, it can cleave additional pepsinogen molecules to produce more pepsin.
Cofactor Binding
Certain zymogens require the binding of specific cofactors or ions to become active.
pH-Dependent Activation
Some zymogens are activated by changes in pH, which can cause conformational changes that expose the active site.
Major Examples of Zymogens
1. Digestive Enzyme Zymogens
The digestive system provides the most well-known examples of zymogens. These are produced by the pancreas and stomach to break down food components.
#### Pancreatic Zymogens
Trypsinogen
Activated to: Trypsin
Activation mechanism: Cleavage by enterokinase in the duodenum
Function: Cleaves proteins at basic amino acids (lysine, arginine)
Clinical significance: Key initiator of pancreatic enzyme activation cascade
Chymotrypsinogen
Activated to: Chymotrypsin
Activation mechanism: Cleavage by trypsin
Function: Cleaves proteins at aromatic amino acids (phenylalanine, tyrosine, tryptophan)
Proelastase
Activated to: Elastase
Activation mechanism: Cleavage by trypsin
Function: Cleaves elastin and other proteins at small, uncharged amino acids
Procarboxypeptidase A and B
Activated to: Carboxypeptidase A and B
Activation mechanism: Cleavage by trypsin
Function: Remove amino acids from the carboxyl terminus of proteins
#### Gastric Zymogens
Pepsinogen
Activated to: Pepsin
Activation mechanism: Acid-catalyzed cleavage and autocatalysis
Function: Initiates protein digestion in the acidic stomach environment
Clinical significance: Pepsinogen levels are used as biomarkers for gastric health
2. Blood Coagulation Zymogens
The blood clotting cascade involves numerous zymogens that are sequentially activated to form blood clots. This system demonstrates the importance of precise regulatory control.
Factor XII (Hageman Factor)
Activated to: Factor XIIa
Function: Initiates intrinsic pathway of coagulation
Factor XI
Activated to: Factor XIa
Function: Activates Factor IX
Factor X
Activated to: Factor Xa
Function: Central component of the common pathway
Prothrombin (Factor II)
Activated to: Thrombin
Function: Converts fibrinogen to fibrin, forming clot structure
Fibrinogen
Activated to: Fibrin
Function: Forms the structural framework of blood clots
3. Complement System Zymogens
The complement system, part of innate immunity, relies heavily on zymogen activation cascades.
C3
Activated to: C3a and C3b
Function: Central complement component; C3b opsonizes pathogens
C5
Activated to: C5a and C5b
Function: C5a is a potent inflammatory mediator; C5b initiates membrane attack complex formation
4. Apoptosis-Related Zymogens
Programmed cell death involves activation of caspase zymogens.
Procaspases
Activated to: Caspases
Function: Execute apoptotic cell death
Examples: Procaspase-8, procaspase-9, procaspase-3
Clinical Significance of Zymogens
Understanding zymogens is crucial for medical practice and diagnosis:
Pancreatitis
Premature activation of pancreatic zymogens within the pancreas leads to acute pancreatitis. This occurs when protective mechanisms fail, causing autodigestion of pancreatic tissue.
Key points for medical exams:
Trypsinogen activation is the initiating event
Can be triggered by gallstones, alcohol, or trauma
Results in severe abdominal pain and elevated pancreatic enzymes
Peptic Ulcer Disease
Dysregulation of pepsinogen activation contributes to peptic ulcer formation. Increased pepsin activity can damage the gastric and duodenal mucosa.
Coagulation Disorders
Many bleeding disorders result from defects in coagulation factor zymogens:
Hemophilia A: Factor VIII deficiency
Hemophilia B: Factor IX deficiency
Warfarin therapy: Inhibits vitamin K-dependent zymogen synthesis
Diagnostic Applications
Zymogen levels serve as important clinical biomarkers:
Pepsinogen I/II ratio: Gastric cancer and H. pylori screening
Pancreatic enzyme levels: Pancreatic function assessment
Coagulation factor levels: Bleeding disorder diagnosis
Zymogen Regulation and Control
The activation of zymogens is tightly regulated through multiple mechanisms:
Inhibitory Proteins
Many systems include specific inhibitors that prevent inappropriate zymogen activation:
Pancreatic secretory trypsin inhibitor (PSTI): Prevents premature trypsin activation
Antithrombin III: Regulates coagulation cascade
C1 esterase inhibitor: Controls complement activation
Compartmentalization
Zymogens are often stored in specific cellular compartments or secreted to particular locations where activation is appropriate.
Enzyme Cascades
Many zymogen systems involve cascading activation, where one activated enzyme activates multiple downstream zymogens, providing signal amplification.
Study Tips for Medical Students
When studying zymogens for medical exams, focus on these key areas:
1. Memorize Major Examples
Create flashcards linking each zymogen to its active form, activation mechanism, and function. Practice with biochemistry flashcards to reinforce your knowledge.
2. Understand Clinical Correlations
Connect zymogen dysfunction to specific diseases. This knowledge is frequently tested in clinical scenarios.
3. Focus on Regulatory Mechanisms
Understand how zymogen activation is controlled, as this often appears in exam questions about enzyme regulation.
4. Practice Pathway Questions
Work through enzyme cascade problems, particularly for coagulation and complement pathways. Use biochemistry practice questions to test your understanding.
5. Review Protein Structure Concepts
Since zymogens involve protein conformational changes, review protein structure and function concepts.
Common Exam Questions About Zymogens
Medical entrance exams frequently test zymogen knowledge through various question types:
Direct Definition Questions
"What is the inactive form of pepsin called?"
"Which enzyme activates trypsinogen?"
Clinical Scenario Questions
"A patient with acute pancreatitis shows elevated enzymes. What is the underlying mechanism?"
"Why don't pancreatic enzymes digest the pancreas under normal conditions?"
Mechanism Questions
"Describe the activation cascade of pancreatic enzymes."
"How does pH affect pepsinogen activation?"
Comparative Questions
"Compare the activation mechanisms of trypsinogen and pepsinogen."
"Which zymogens are involved in the intrinsic pathway of coagulation?"
Future Directions and Research
Current research in zymogen biology focuses on:
Therapeutic Applications
Targeted cancer therapy: Activating apoptotic zymogens selectively in cancer cells
Anticoagulant development: Creating specific inhibitors of coagulation zymogens
Digestive disorders: Developing enzyme replacement therapies
Diagnostic Advances
Biomarker development: Using zymogen levels for early disease detection
Point-of-care testing: Rapid assessment of enzyme activation status
Drug Development
Understanding zymogen activation mechanisms helps in designing drugs that can modulate these pathways for therapeutic benefit.
Conclusion
Zymogens represent a sophisticated biological control mechanism that allows precise regulation of enzyme activity. From digestion to blood clotting to immune responses, these enzyme precursors are fundamental to human physiology. For medical students, mastering zymogen concepts is essential for understanding biochemistry, physiology, and pathology.
The key to success in learning about zymogens is understanding both the molecular mechanisms and clinical applications. Focus on the major examples, memorize the activation pathways, and always consider the clinical significance of zymogen dysfunction.
Remember that zymogens demonstrate the elegant solutions evolution has developed to control potentially dangerous enzymatic processes. By storing enzymes in inactive forms and activating them only when and where needed, our bodies maintain precise control over critical biological processes.
To deepen your understanding of enzyme function and regulation, explore comprehensive biochemistry lessons and test your knowledge with targeted practice questions. Understanding zymogens will provide a strong foundation for more advanced topics in medical biochemistry and clinical medicine.