Enzymes

On this page
🧬 Enzyme Architecture: The Molecular Machinery Mastery
Enzymes are the master catalysts that make life possible, accelerating reactions by factors of millions while maintaining exquisite specificity. You'll discover how their three-dimensional architecture creates active sites that stabilize transition states, how kinetic principles predict their behavior under varying conditions, and how cells regulate these molecular machines through feedback and allosteric control. We'll connect enzyme mechanisms to clinical reality-from diagnosing myocardial infarction through troponin and CK-MB to understanding drug targets and inborn errors of metabolism. By integrating structure, kinetics, regulation, and pathology, you'll command the enzymatic principles that govern both normal physiology and disease.
📌 Remember: PLEASE - Protein catalysts, Lower activation energy, Enzyme-substrate complex, Active site specificity, Speed up reactions, Energy barriers reduced. Every enzyme follows these six fundamental principles with 10^6 to 10^17-fold rate enhancement over uncatalyzed reactions.
Enzyme Structural Hierarchy: From Atoms to Function
- Primary Structure (20 standard amino acids)
- Linear sequence determines all higher-order structure
- Single amino acid changes can reduce activity by >95%
- Sickle cell: Glu→Val substitution in β-globin
- Phenylketonuria: Phe hydroxylase mutations (>400 variants)
- Secondary Structure (α-helices and β-sheets)
- 60-70% of enzyme structure in organized motifs
- Critical for active site geometry and stability
- Tertiary Structure (3D folding pattern)
- Creates active site with Å-level precision
- Binding pocket complementary to transition state
- Substrate affinity: μM to mM range
- Transition state affinity: 10^6-fold higher
- Quaternary Structure (multi-subunit assembly)
- 40% of enzymes exist as oligomers
- Enables allosteric regulation and cooperativity
| Structural Level | Key Features | Clinical Significance | Stability Factors | Disruption Effects |
|---|---|---|---|---|
| Primary | Amino acid sequence | Genetic mutations | Peptide bonds | Complete loss of function |
| Secondary | α-helices, β-sheets | Protein folding diseases | Hydrogen bonds | Partial activity loss |
| Tertiary | 3D active site | Drug binding sites | Multiple weak forces | Variable activity changes |
| Quaternary | Subunit assembly | Allosteric regulation | Subunit interactions | Regulatory dysfunction |

Active Site Architecture: Precision Engineering
The active site represents molecular engineering at its finest, creating an environment that stabilizes transition states while destabilizing ground states. This 10-20 amino acid region determines substrate specificity and catalytic efficiency.
- Binding Subsites (substrate recognition)
- S1, S2, S3 subsites accommodate specific residues
- Lock-and-key vs induced-fit models
- Rigid complementarity: 10^3-fold specificity
- Conformational adaptation: 10^6-fold rate enhancement
- Catalytic Residues (chemical transformation)
- His, Asp, Glu, Cys, Ser most common catalytic residues
- Proton donors/acceptors with pKa values 6-8
- Optimal activity near physiological pH (7.4)
- ±1 pH unit change reduces activity by 50-90%
💡 Master This: Active site geometry explains drug selectivity. Aspirin acetylates Ser530 in COX-1 and Ser516 in COX-2, but 200-fold selectivity differences arise from active site volume variations (25% larger in COX-2).
Understanding enzyme architecture provides the foundation for comprehending how these molecular machines achieve their remarkable catalytic power through precise structural organization.
🧬 Enzyme Architecture: The Molecular Machinery Mastery
⚡ Catalytic Mechanisms: The Reaction Acceleration Engine
📌 Remember: FASTER - Facilitate transition states, Activation energy lowered, Stabilize intermediates, Temperature independence, Energy barriers reduced, Reaction rates enhanced. Enzymes achieve 10^8-fold rate acceleration through transition state stabilization alone.
Catalytic Strategies: The Molecular Toolkit
- Acid-Base Catalysis (proton transfer mechanisms)
- General acid: Proton donation to leaving groups
- General base: Proton abstraction from nucleophiles
- His57 in serine proteases: pKa 6.0
- Asp102 carboxyl group: pKa 3.9
- Optimal activity when pKa matches reaction pH
- Covalent Catalysis (transient bond formation)
- Nucleophilic attack by enzyme residues
- Acyl-enzyme intermediates in serine proteases
- Ser195 forms covalent bond with substrate
- Intermediate lifetime: milliseconds
- 1000-fold rate enhancement over non-covalent
- Metal Ion Catalysis (electrophilic assistance)
- Lewis acid activation of electrophiles
- Redox reactions through metal oxidation states
- Zn^2+: >300 enzymes require zinc
- Mg^2+: >600 enzymes, especially kinases
- Fe^2+/Fe^3+: Cytochrome oxidase, catalase
| Catalytic Strategy | Mechanism | Rate Enhancement | Clinical Examples | Metal Requirements |
|---|---|---|---|---|
| Acid-Base | Proton transfer | 10^2-10^4 fold | Pepsin, trypsin | None |
| Covalent | Transient bonds | 10^3-10^5 fold | Chymotrypsin, acetylcholinesterase | None |
| Metal Ion | Electrophilic activation | 10^4-10^6 fold | Carbonic anhydrase, alcohol dehydrogenase | Zn^2+, Mg^2+ |
| Electrostatic | Charge stabilization | 10^2-10^3 fold | Triose phosphate isomerase | None |
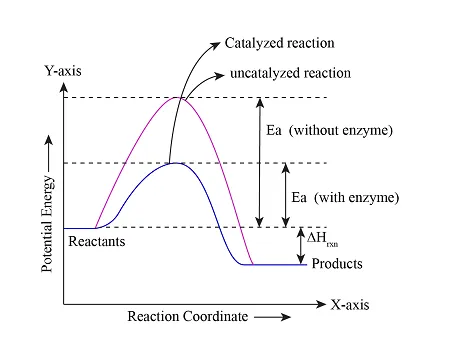
Transition State Theory: The Energy Landscape
Enzymes achieve their remarkable catalytic power by preferentially binding and stabilizing transition states rather than ground-state substrates. This fundamental principle explains both catalytic efficiency and inhibitor design.
- Transition State Stabilization (primary mechanism)
- 10^6-fold higher affinity for transition state vs substrate
- ΔG‡ reduction of 5-23 kcal/mol
- Each 1.4 kcal/mol reduction = 10-fold rate increase
- Room temperature: kT = 0.6 kcal/mol
- Substrate Destabilization (ground state effects)
- Induced fit creates strain in substrate bonds
- Conformational selection favors reactive conformers
- Enthalpy cost: 2-5 kcal/mol substrate distortion
- Entropy gain: Reduced conformational freedom
💡 Master This: Transition state analogs make the most potent inhibitors. Statins mimic the transition state of HMG-CoA reductase with nM binding affinity (10^6-fold tighter than substrate), explaining their exceptional potency in cholesterol reduction.
Understanding catalytic mechanisms reveals how enzymes achieve their extraordinary efficiency and provides the foundation for rational drug design and metabolic pathway analysis.
⚡ Catalytic Mechanisms: The Reaction Acceleration Engine
🎯 Enzyme Kinetics: The Velocity Command Center
📌 Remember: KINETIC - Km is half-maximal velocity, Initial rates measured, Non-competitive changes Vmax, Enzyme concentration affects Vmax, Temperature increases rates, Inhibitors alter patterns, Competitive inhibition increases Km. These seven principles govern all enzyme kinetic behavior.
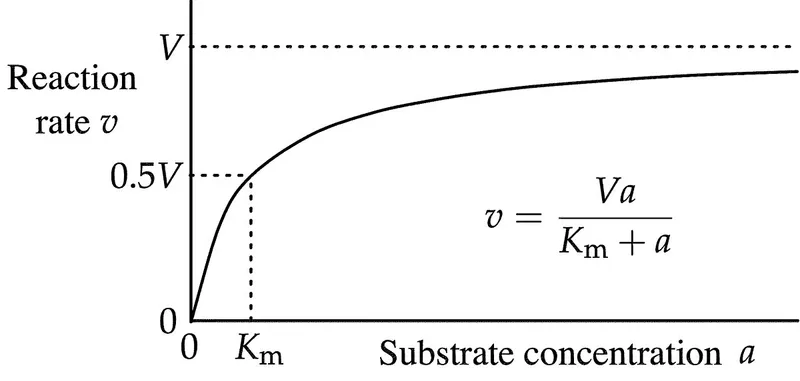
Michaelis-Menten Fundamentals: The Velocity Equation
The Michaelis-Menten equation describes enzyme kinetics through two fundamental parameters that determine reaction rates and clinical significance.
- Vmax (maximum velocity)
- Achieved at saturating substrate concentrations
- Directly proportional to enzyme concentration
- Vmax = kcat × [E]total
- kcat (turnover number): 1-10^6 s^-1
- Catalase: 4 × 10^7 s^-1 (fastest known)
- Km (Michaelis constant)
- Substrate concentration at ½ Vmax
- Inverse measure of enzyme-substrate affinity
- Low Km (μM): High affinity, efficient at low [S]
- High Km (mM): Low affinity, requires high [S]
- Physiological [S] typically 0.1-10 × Km
$$v = \frac{V_{max}[S]}{K_m + [S]}$$
| Kinetic Parameter | Typical Range | Clinical Significance | Measurement Units | Physiological Impact |
|---|---|---|---|---|
| Vmax | 10^-9 to 10^-3 M/s | Enzyme concentration/activity | mol/L/s | Metabolic flux capacity |
| Km | 10^-6 to 10^-2 M | Substrate affinity | mol/L | Sensitivity to substrate |
| kcat | 1 to 10^7 s^-1 | Catalytic efficiency | s^-1 | Turnover rate |
| kcat/Km | 10^3 to 10^8 M^-1s^-1 | Overall efficiency | M^-1s^-1 | Physiological effectiveness |

Inhibition Patterns: Kinetic Fingerprints
Different inhibition mechanisms produce characteristic kinetic patterns that enable inhibitor classification and drug mechanism determination.
- Competitive Inhibition (active site competition)
- Increased Km, unchanged Vmax
- Reversible by increasing substrate concentration
- Ki (inhibitor constant): nM to μM for drugs
- α = 1 + [I]/Ki (apparent Km increase)
- Non-competitive Inhibition (allosteric binding)
- Unchanged Km, decreased Vmax
- Cannot be overcome by substrate increase
- Mixed inhibition: Both Km and Vmax affected
- Uncompetitive: Decreased Km and Vmax
- Irreversible Inhibition (covalent modification)
- Time-dependent inactivation
- Pseudo-first-order kinetics at fixed [I]
- Aspirin: t½ = 30 minutes for COX acetylation
- Organophosphates: Hours to days for recovery
💡 Master This: Inhibition patterns guide therapeutic strategies. Statins show competitive inhibition of HMG-CoA reductase (Ki = 1-50 nM), requiring evening dosing when endogenous HMG-CoA synthesis peaks, maximizing therapeutic efficiency.
Understanding enzyme kinetics provides quantitative frameworks for predicting drug responses, optimizing dosing regimens, and interpreting clinical laboratory results with mathematical precision.
🎯 Enzyme Kinetics: The Velocity Command Center
🔬 Enzyme Regulation: The Metabolic Control Matrix
📌 Remember: REGULATE - Reversible modifications, Effector binding sites, Glycogen phosphorylase example, Upstream signal integration, Long-range conformational changes, Allosteric cooperativity, Time-dependent responses, Energy state sensing. These eight mechanisms enable precise metabolic control.
Allosteric Regulation: The Conformational Switch Network
Allosteric regulation enables enzymes to sense cellular energy states and respond to regulatory signals through conformational changes that alter catalytic activity without affecting the active site directly.
- Positive Allosteric Effectors (activity enhancers)
- AMP activates phosphofructokinase (PFK)
- Ca^2+ activates pyruvate dehydrogenase
- 10-fold activity increase at μM concentrations
- Sigmoidal kinetics indicate cooperativity
- Hill coefficient >1 demonstrates positive cooperativity
- Negative Allosteric Effectors (activity inhibitors)
- ATP inhibits phosphofructokinase (IC50 = 0.5 mM)
- Citrate inhibits acetyl-CoA carboxylase
- Feedback inhibition prevents overproduction
- >90% inhibition at 2-5 mM concentrations
- Heterotropic Effects (different ligand binding)
- Effector binding affects substrate affinity
- Concerted model: All subunits change together
- T-state (tense): Low substrate affinity
- R-state (relaxed): High substrate affinity
| Regulatory Enzyme | Positive Effectors | Negative Effectors | Physiological Role | Clinical Significance |
|---|---|---|---|---|
| Phosphofructokinase | AMP, ADP, Pi | ATP, citrate | Glycolytic flux control | Diabetes, cancer metabolism |
| Pyruvate kinase | Fructose-1,6-BP | ATP, alanine | Glycolytic commitment | Hemolytic anemia |
| Acetyl-CoA carboxylase | Citrate, insulin | Palmitoyl-CoA, glucagon | Fatty acid synthesis | Obesity, metabolic syndrome |
| Glutamine synthetase | α-Ketoglutarate | Glycine, histidine | Nitrogen metabolism | Hepatic encephalopathy |
Covalent Modification: The Molecular Switch Network
Covalent modifications provide rapid, reversible enzyme regulation through post-translational modifications that alter enzyme activity, localization, and protein interactions.
- Phosphorylation/Dephosphorylation (most common)
- Protein kinases: >500 human enzymes
- Protein phosphatases: >150 human enzymes
- Ser/Thr kinases: 90% of phosphorylation sites
- Tyr kinases: 10% but crucial for signaling
- Phosphorylation can activate or inhibit
- Acetylation/Deacetylation (metabolic regulation)
- Lysine acetylation affects >2000 proteins
- Sirtuins (NAD+-dependent deacetylases)
- SIRT1 regulates >100 metabolic enzymes
- Caloric restriction increases sirtuin activity
- Ubiquitination (protein degradation)
- Polyubiquitin targets proteins for 26S proteasome
- Half-life regulation: minutes to days
- Cyclins: 30-60 minutes during cell cycle
- p53: 20 minutes under normal conditions
💡 Master This: Phosphorylation cascades amplify signals exponentially. Epinephrine binding to β-adrenergic receptors activates adenylyl cyclase, producing cAMP that activates protein kinase A, which phosphorylates phosphorylase kinase, ultimately activating glycogen phosphorylase with >1000-fold signal amplification.
Understanding enzyme regulation reveals how cells maintain metabolic homeostasis and respond to physiological challenges through sophisticated control networks that integrate multiple regulatory signals.
🔬 Enzyme Regulation: The Metabolic Control Matrix
🏥 Clinical Enzymology: The Diagnostic Powerhouse
📌 Remember: CARDIAC - CK-MB rises first (4-6 hours), AST peaks early (24-48 hours), Rise patterns indicate timing, Duration varies by enzyme, Isoenzymes provide specificity, Activity correlates with damage, Clearance determines duration. These seven principles guide cardiac enzyme interpretation.
Cardiac Biomarkers: The Heart Attack Timeline
Cardiac enzyme patterns provide precise timing and extent of myocardial damage through characteristic release patterns and tissue specificity.
- Troponin I/T (gold standard markers)
- Rises: 3-4 hours post-MI
- Peaks: 12-24 hours
- Duration: 7-14 days
- Sensitivity: >95% at 6 hours
- Specificity: >90% for myocardial damage
- Detection limit: 0.01-0.04 ng/mL
- CK-MB (creatine kinase muscle-brain)
- Rises: 4-6 hours post-MI
- Peaks: 12-24 hours
- Duration: 2-3 days
- Normal: <6.3 ng/mL (males), <4.3 ng/mL (females)
- CK-MB/Total CK ratio: >2.5% suggests cardiac origin
- Myoglobin (early marker)
- Rises: 1-3 hours post-MI
- Peaks: 6-12 hours
- Duration: 24-36 hours
- High sensitivity but low specificity
- Useful for early detection and reperfusion monitoring
| Cardiac Marker | Rise Time | Peak Time | Duration | Sensitivity | Specificity | Clinical Use |
|---|---|---|---|---|---|---|
| Troponin I/T | 3-4 hours | 12-24 hours | 7-14 days | >95% | >90% | Diagnosis, prognosis |
| CK-MB | 4-6 hours | 12-24 hours | 2-3 days | 85-95% | 80-90% | Timing, reinfarction |
| Myoglobin | 1-3 hours | 6-12 hours | 24-36 hours | >95% | <50% | Early detection |
| LDH | 12-24 hours | 48-72 hours | 7-10 days | 80-85% | <60% | Late marker |
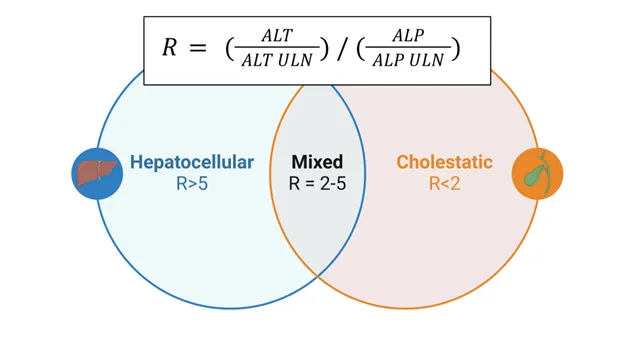
Hepatic Enzymes: The Liver Function Matrix
Liver enzyme patterns distinguish between hepatocellular injury, cholestatic disease, and mixed patterns through characteristic elevation ratios and temporal profiles.
- Aminotransferases (hepatocellular markers)
- ALT (alanine aminotransferase): Liver-specific
- AST (aspartate aminotransferase): Multi-organ
- Normal ALT: <40 U/L (males), <32 U/L (females)
- Normal AST: <40 U/L (both sexes)
- AST/ALT ratio: <1 in viral hepatitis, >2 in alcoholic liver disease
- Cholestatic Enzymes (biliary obstruction markers)
- Alkaline phosphatase (ALP): Normal <120 U/L
- γ-Glutamyl transferase (GGT): Normal <60 U/L
- ALP elevation >3× normal: Suggests cholestasis
- GGT elevation confirms hepatic origin of ALP
- ALP/GGT ratio: Helps distinguish bone vs liver disease
💡 Master This: Pattern recognition guides diagnosis. ALT >1000 U/L with AST/ALT <1 suggests acute viral hepatitis or drug-induced liver injury, while AST/ALT >2 with GGT >100 U/L indicates alcoholic hepatitis with >80% specificity.
Understanding clinical enzymology transforms laboratory data into precise diagnostic tools that enable accurate disease detection, treatment monitoring, and prognostic assessment.
🏥 Clinical Enzymology: The Diagnostic Powerhouse
🔗 Enzyme Integration: The Metabolic Network Architecture
📌 Remember: NETWORK - Network effects amplify signals, Energy charge regulates pathways, Tissue-specific expression patterns, Whole-body coordination, Opposing pathways reciprocally regulated, Redox state influences activity, Key enzymes control flux. These seven principles govern metabolic integration.
Metabolic Pathway Coordination: The Flux Control Network
Metabolic pathways operate as integrated networks where rate-limiting enzymes control flux and regulatory circuits coordinate opposing pathways to maintain homeostasis.
- Glycolysis-Gluconeogenesis Coordination
- Reciprocal regulation prevents futile cycling
- Key regulatory enzymes: PFK-1/F-1,6-BPase, PK/PEPCK
- Fructose-2,6-bisphosphate: Allosteric coordinator
- Fed state: High F-2,6-BP activates glycolysis
- Fasting state: Low F-2,6-BP activates gluconeogenesis
- Hormonal control: Insulin vs glucagon signaling
- Fatty Acid Synthesis-Oxidation Balance
- Acetyl-CoA carboxylase (synthesis) vs CPT-1 (oxidation)
- Malonyl-CoA coordinates both pathways
- Synthesis: Malonyl-CoA required substrate
- Oxidation: Malonyl-CoA inhibits CPT-1
- >95% inhibition of β-oxidation during lipogenesis
| Metabolic State | Dominant Pathways | Key Regulators | Enzyme Activities | Clinical Markers |
|---|---|---|---|---|
| Fed State | Glycolysis, lipogenesis | Insulin, F-2,6-BP | PFK↑, ACC↑, CPT-1↓ | Glucose ↓, insulin ↑ |
| Fasting State | Gluconeogenesis, β-oxidation | Glucagon, cortisol | PEPCK↑, CPT-1↑, ACC↓ | Glucose ↑, ketones ↑ |
| Exercise | Glycolysis, fatty acid oxidation | Epinephrine, AMP | PFK↑, CPT-1↑, PDH↑ | Lactate ↑, FFA ↑ |
| Diabetes | Gluconeogenesis, ketogenesis | Insulin deficiency | PEPCK↑↑, HMG-CoA synthase↑ | Glucose ↑↑, ketones ↑↑ |

Tissue-Specific Metabolic Specialization: The Organ Network
Different tissues express unique enzyme profiles that enable metabolic specialization and inter-organ cooperation to maintain whole-body homeostasis.
- Liver (metabolic hub)
- Glucokinase: High Km (10 mM) glucose sensor
- PEPCK: Gluconeogenesis capacity
- Glucose buffer: Takes up glucose when >5 mM
- Glucose production: 2-3 mg/kg/min during fasting
- Urea cycle: >95% of ammonia detoxification
- Muscle (glucose utilization and storage)
- Hexokinase: Low Km (0.1 mM) for glucose uptake
- Glycogen phosphorylase: Rapid energy mobilization
- Glucose uptake: >80% of postprandial glucose
- Glycogen storage: 300-500 g in trained individuals
- Brain (glucose-dependent metabolism)
- High-affinity glucose transporters (GLUT-1, GLUT-3)
- Glucose consumption: 120-140 g/day (20% of total)
- Ketone utilization: Up to 60% during prolonged fasting
- Blood-brain barrier: Selective enzyme expression
💡 Master This: Tissue enzyme patterns explain drug distribution. Statins concentrate in liver due to OATP1B1 transporters and high HMG-CoA reductase expression, achieving >10-fold higher concentrations than plasma, explaining hepatic selectivity and muscle-sparing effects at therapeutic doses.
Understanding enzyme network integration reveals how metabolic pathways coordinate to maintain homeostasis and how disruptions lead to disease states requiring targeted therapeutic interventions.
🔗 Enzyme Integration: The Metabolic Network Architecture
🎯 Enzyme Mastery: The Clinical Command Arsenal
📌 Remember: MASTERY - Multiple enzyme patterns, Assess timing and trends, Specificity guides diagnosis, Tissue sources matter, Elevation magnitude significant, Ratio analysis crucial, Yield increases with integration. Master these seven principles for clinical excellence.
Essential Enzyme Reference Arsenal
| Clinical Scenario | Key Enzymes | Critical Values | Diagnostic Patterns | Time Course |
|---|---|---|---|---|
| Acute MI | Troponin I/T, CK-MB | >0.04 ng/mL, >6.3 ng/mL | Troponin most specific | 3-4h rise, 7-14d duration |
| Hepatitis | ALT, AST | >10× normal | ALT > AST, AST/ALT <1 | Peak 1-3 days |
| Cholestasis | ALP, GGT | ALP >3× normal | ALP ↑↑, GGT confirms hepatic | Gradual rise |
| Pancreatitis | Lipase, Amylase | >3× normal | Lipase more specific | 6-12h rise, 3-5d duration |
| Muscle Disease | CK, Aldolase | CK >10× normal | CK-MM predominant | Variable timing |
Rapid Pattern Recognition Framework
- Hepatocellular Pattern (ALT/AST predominant)
- AST/ALT <1: Viral hepatitis, drug toxicity
- AST/ALT >2: Alcoholic hepatitis, cirrhosis
- Both >1000 U/L: Acute hepatic necrosis
- Cholestatic Pattern (ALP/GGT predominant)
- ALP >3× + GGT >2×: Biliary obstruction
- ALP isolated: Consider bone disease
- GGT >5×: Alcohol-related liver disease
- Mixed Pattern (Both elevated)
- Moderate elevation: Drug hepatotoxicity
- Progressive rise: Evolving hepatic injury
💡 Master This: Enzyme ratios provide diagnostic precision. CK-MB/Total CK >2.5% indicates cardiac origin with >90% specificity, while AST/ALT >2 with GGT >100 U/L suggests alcoholic liver disease with >80% positive predictive value in appropriate clinical context.
This clinical command arsenal transforms enzyme knowledge into practical tools for immediate patient assessment, accurate diagnosis, and evidence-based treatment decisions that optimize clinical outcomes.
🎯 Enzyme Mastery: The Clinical Command Arsenal
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more
Have doubts about this lesson?
Ask Rezzy, your AI Study Mate, to explain anything you didn't understand
Everything you need for NEET-PG prep
Get full Oncourse access with lessons, practice questions, flashcards and AI study tools.
Scan to download app
























