Gram-negative — MCQs

On this page
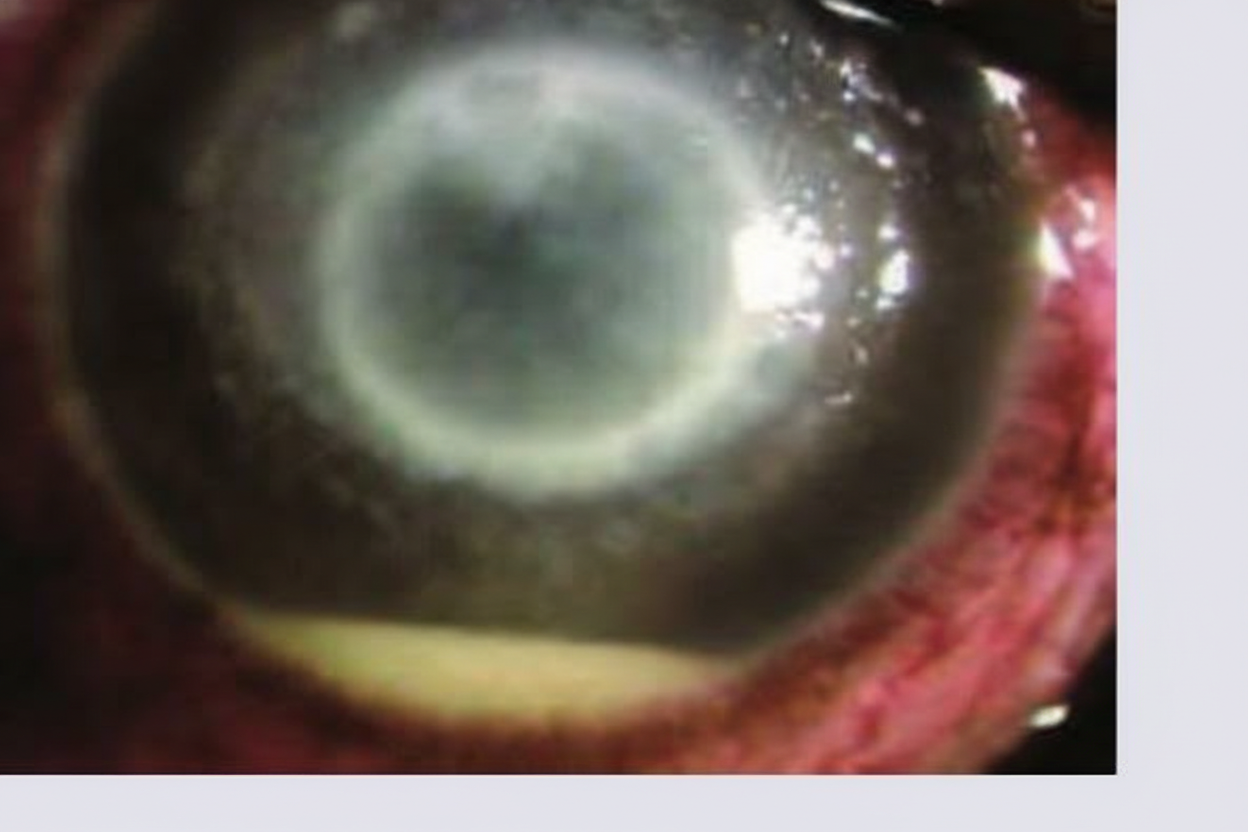
A 45-year-old man comes to the physician because of a 1-day history of progressive pain and blurry vision of his right eye. He has difficulties opening the eye because of pain. His left eye is asymptomatic. He wears contact lenses. He has bronchial asthma treated with inhaled salbutamol. He works as a kindergarten teacher. His temperature is 37°C (98.6°F), pulse is 85/min, and blood pressure is 135/75 mm Hg. Examination shows a visual acuity in the left eye of 20/25 and the ability to count fingers at 3 feet in the right eye. A photograph of the right eye is shown. Which of the following is the most likely diagnosis?
A 23-year-old man comes to the physician because of a 2-day history of profuse watery diarrhea and abdominal cramps. Four days ago, he returned from a backpacking trip across Southeast Asia. Physical examination shows dry mucous membranes and decreased skin turgor. Stool culture shows gram-negative, oxidase-positive, curved rods that have a single polar flagellum. The pathogen responsible for this patient's condition most likely has which of the following characteristics?
An 83-year-old male presents to the emergency department with altered mental status. The patient’s vitals signs are as follows: temperature is 100.7 deg F (38.2 deg C), blood pressure is 143/68 mmHg, heart rate is 102/min, and respirations are 22/min. The caretaker states that the patient is usually incontinent of urine, but she has not seen any soiled adult diapers in the past 48 hours. A foley catheter is placed with immediate return of a large volume of cloudy, pink urine. Which of the following correctly explains the expected findings from this patient’s dipstick urinalysis?
A 10-year-old boy is brought in to the emergency room by his parents after he complained of being very weak during a soccer match the same day. The parents noticed that yesterday, the patient seemed somewhat clumsy during soccer practice and was tripping over himself. Today, the patient fell early in his game and complained that he could not get back up. The patient is up-to-date on his vaccinations and has no previous history of illness. The parents do report that the patient had abdominal pain and bloody diarrhea the previous week, but the illness resolved without antibiotics or medical attention. The patient’s temperature is 100.9°F (38.3°C), blood pressure is 110/68 mmHg, pulse is 84/min, and respirations are 14/min. On exam, the patient complains of tingling sensations that seem reduced in his feet. He has no changes in vibration or proprioception. Achilles and patellar reflexes are 1+ bilaterally. On strength testing, foot dorsiflexion and plantar flexion are 3/5 and knee extension and knee flexion are 4-/5. Hip flexion, hip extension, and upper extremity strength are intact. Based on this clinical history and physical exam, what pathogenic agent could have been responsible for the patient’s illness?
A 4-year-old boy is brought to the physician because of a 1-day history of passing small quantities of dark urine. Two weeks ago, he had fever, abdominal pain, and bloody diarrhea for several days that were treated with oral antibiotics. Physical examination shows pale conjunctivae and scleral icterus. His hemoglobin concentration is 7.5 g/dL, platelet count is 95,000/mm3, and serum creatinine concentration is 1.9 mg/dL. A peripheral blood smear shows irregular red blood cell fragments. Avoiding consumption of which of the following foods would have most likely prevented this patient's condition?
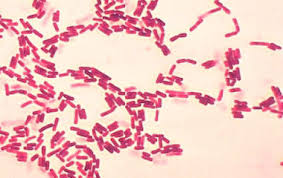
A 43-year-old man hospitalized for acute pancreatitis develops a high-grade fever and productive cough with gelatinous sputum. A sample of his expectorated sputum is obtained and fixed to a microscope slide using heat. A crystal violet dye is applied to the slide, followed by an iodine solution, acetone solution, and lastly, safranin dye. A photomicrograph of the result is shown. Which of the following cell components is responsible for the pink color seen on this stain?
An 86-year-old male with a history of hypertension and hyperlipidemia is sent to the hospital from the skilled nursing facility due to fever, confusion, and decreased urine output. Urinalysis shows 12-18 WBC/hpf with occasional lymphocytes. Urine and blood cultures grow out gram-negative, motile, urease positive rods. What component in the identified bacteria is primarily responsible for causing the innate immune response seen in this patient?
A 46-year-old woman from Ecuador is admitted to the hospital because of tarry-black stools and epigastric pain for 2 weeks. The epigastric pain is relieved after meals, but worsens after 1–2 hours. She has no history of serious illness and takes no medications. Physical examination shows no abnormalities. Fecal occult blood test is positive. Esophagogastroduodenoscopy shows a bleeding duodenal ulcer. Microscopic examination of a duodenal biopsy specimen is most likely to show which of the following?
A 4-year-old boy is brought to the physician because of a 3-day history of fever and left ear pain. Examination of the left ear shows a bulging tympanic membrane with green discharge. Gram stain of the discharge shows a gram-negative coccobacillus. The isolated organism grows on chocolate agar. The causal pathogen most likely produces a virulence factor that acts by which of the following mechanisms?
A 24-year-old man comes to the physician with a 2-day history of fever, crampy abdominal pain, and blood-tinged diarrhea. He recently returned from a trip to Mexico. His temperature is 38.2°C (100.8°F). Abdominal examination shows diffuse tenderness to palpation; bowel sounds are hyperactive. Stool cultures grow nonlactose fermenting, oxidase-negative, gram-negative rods that do not produce hydrogen sulfide on triple sugar iron agar. Which of the following processes is most likely involved in the pathogenesis of this patient's condition?
Practice by Chapter
Neisseria species (meningitidis, gonorrhoeae)
Practice Questions
Escherichia coli
Practice Questions
Klebsiella species
Practice Questions
Proteus species
Practice Questions
Pseudomonas aeruginosa
Practice Questions
Haemophilus influenzae
Practice Questions
Bordetella pertussis
Practice Questions
Legionella pneumophila
Practice Questions
Bacteroides and anaerobic gram-negatives
Practice Questions
Helicobacter pylori
Practice Questions
Campylobacter jejuni
Practice Questions
Vibrio species
Practice Questions
ESBL and CRE pathogens
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
