Arrhythmias — MCQs

On this page
Match the following A. Atrial fibrillation B. Atrial flutter C. PSVT D. Ventricular tachycardia
A patient with a history of hypertension presents with atrial fibrillation, shortness of breath, and bilateral basal crackles on auscultation. Which of the following would be the least important in the management of this patient?
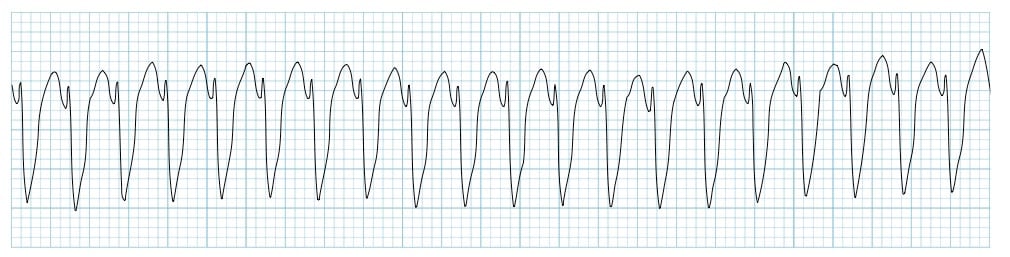
A patient is pulseless with the following rhythm shown in the ECG. What is the next best step in management?
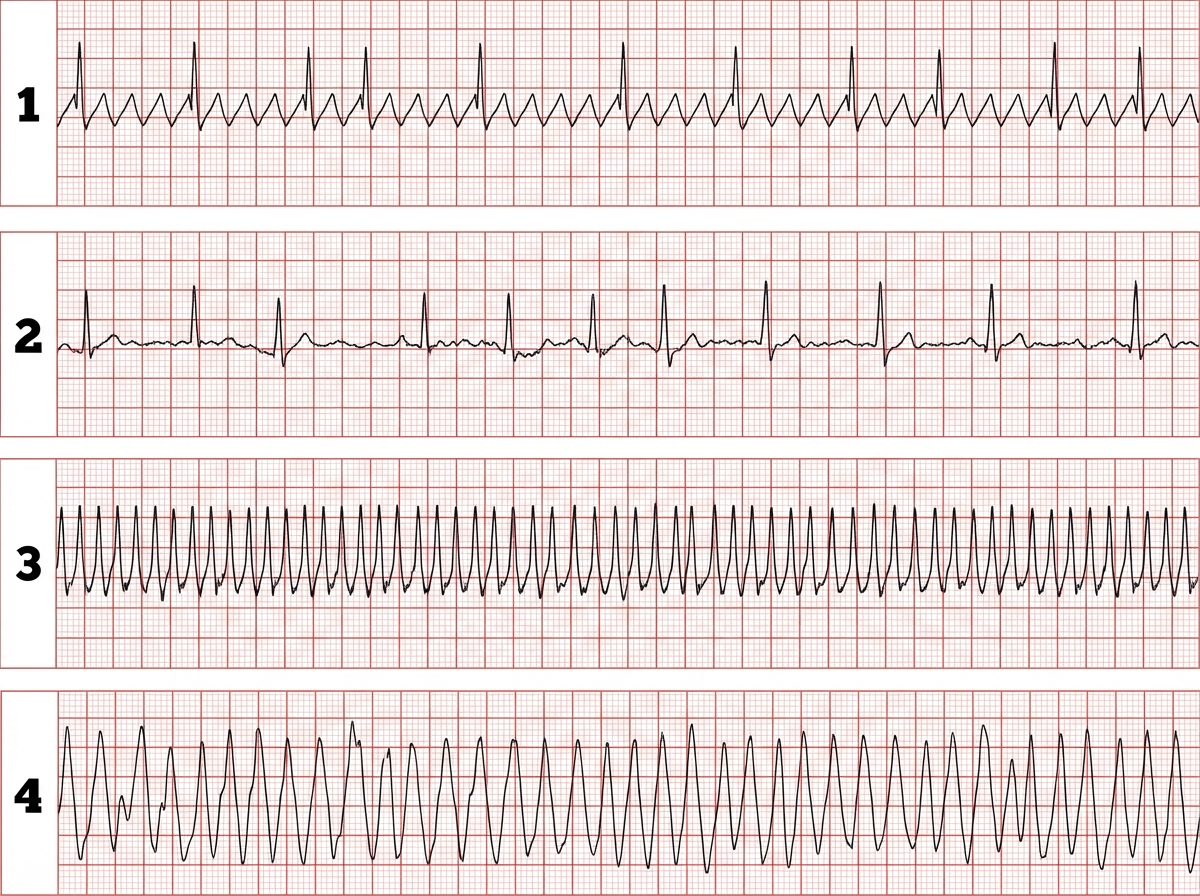
Match the following ECG findings (1-4) with their corresponding arrhythmias (A-D): 1. Sawtooth pattern in leads II, III, aVF with regular ventricular response 2. Irregularly irregular rhythm with absent P waves 3. Narrow QRS tachycardia with abrupt onset/termination 4. Wide QRS tachycardia with AV dissociation A. Atrial fibrillation B. PSVT (Paroxysmal Supraventricular Tachycardia) C. Atrial flutter D. Ventricular tachycardia What is the correct matching?
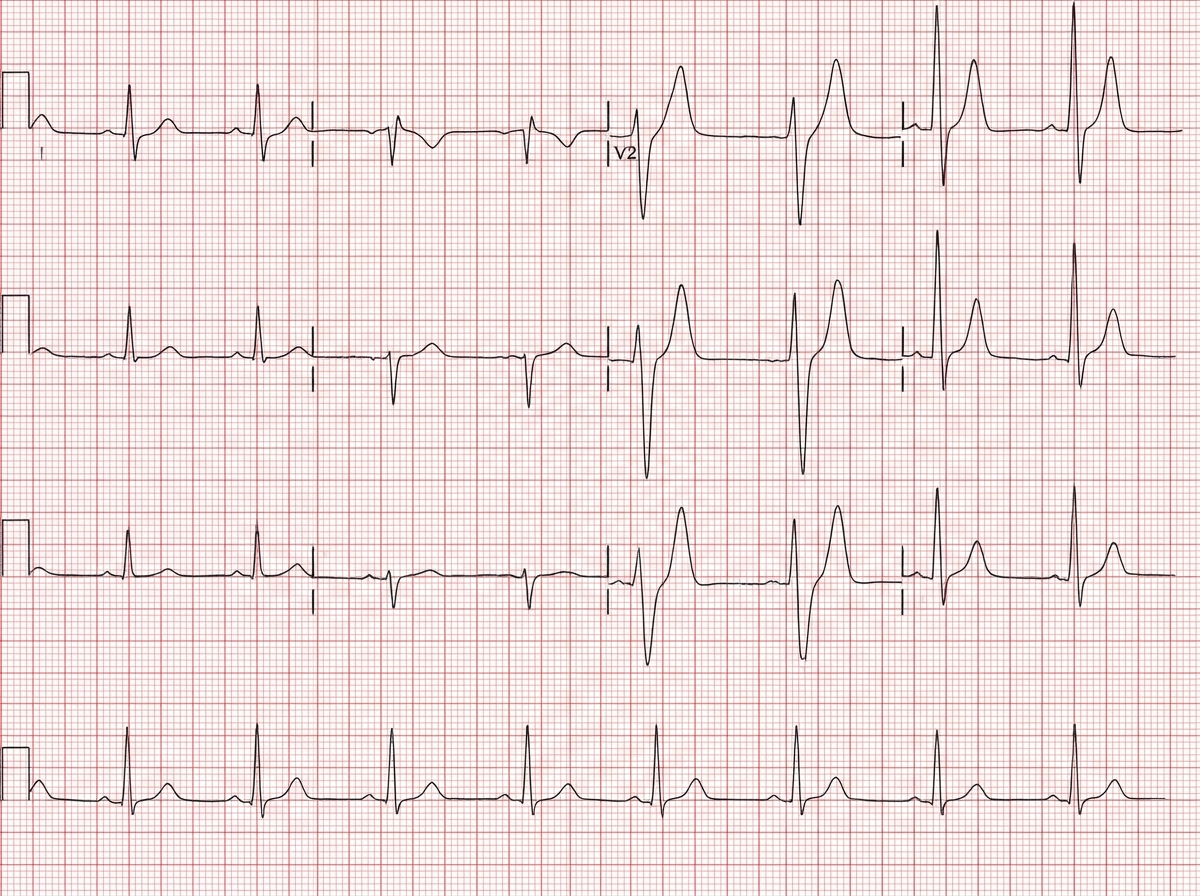
A 45-year-old male is brought into the emergency department by emergency medical services. The patient has a history of substance abuse and was found down in his apartment lying on his right arm. He was last seen 24 hours earlier by his mother who lives in the same building. He is disoriented and unable to answer any questions. His vitals are HR 48, T 97.6, RR 18, BP 100/75. You decide to obtain an EKG as shown in Figure 1. Which of the following is most likely the cause of this patient's EKG results?
Practice by Chapter
Basic electrophysiology
Practice Questions
ECG interpretation fundamentals
Practice Questions
Supraventricular tachycardias
Practice Questions
Atrial fibrillation and flutter
Practice Questions
Ventricular arrhythmias
Practice Questions
Bradyarrhythmias and conduction disorders
Practice Questions
Sudden cardiac death
Practice Questions
Antiarrhythmic medications
Practice Questions
Cardioversion and defibrillation
Practice Questions
Pacemakers and ICDs
Practice Questions
Catheter ablation procedures
Practice Questions
Secondary arrhythmias (electrolyte, drug-induced)
Practice Questions
Inherited arrhythmia syndromes
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
