ACS — MCQs

On this page
A 56-year-old patient developed excruciating chest discomfort in the past 72 hours, relieved by GTN spray. Troponin I is normal, and the ECG shows features of left ventricular hypertrophy (LVH) with T wave flattening. The patient is already on statins, aspirin, and metoprolol 50 mg . What is the next best step in management?
A 78-year-old male with a 35-pack-year smoking history, hyperlipidemia, and peripheral vascular disease is at home eating dinner with his wife when he suddenly has acute onset, crushing chest pain. He lives in a remote rural area, and, by the time the paramedics arrive 30 minutes later, he is pronounced dead. What is the most likely cause of this patient's death?
A 57-year-old man is brought to the emergency department after having chest pain for the last hour. He rates his pain as 8/10, dull in character, and says it is associated with sweating and shortness of breath. He has a history of diabetes and hypercholesterolemia. His current medication list includes amlodipine, aspirin, atorvastatin, insulin, and esomeprazole. He has smoked 2 packs of cigarettes per day for the past 25 years. His blood pressure is 98/66 mm Hg, pulse is 110/min, oxygen saturation is 94% on room air, and BMI is 31.8 kg/m2. His lungs are clear to auscultation. An electrocardiogram (ECG) is shown below. The patient is given 325 mg of oral aspirin and sublingual nitroglycerin. What is the most appropriate next step in the management of this condition?
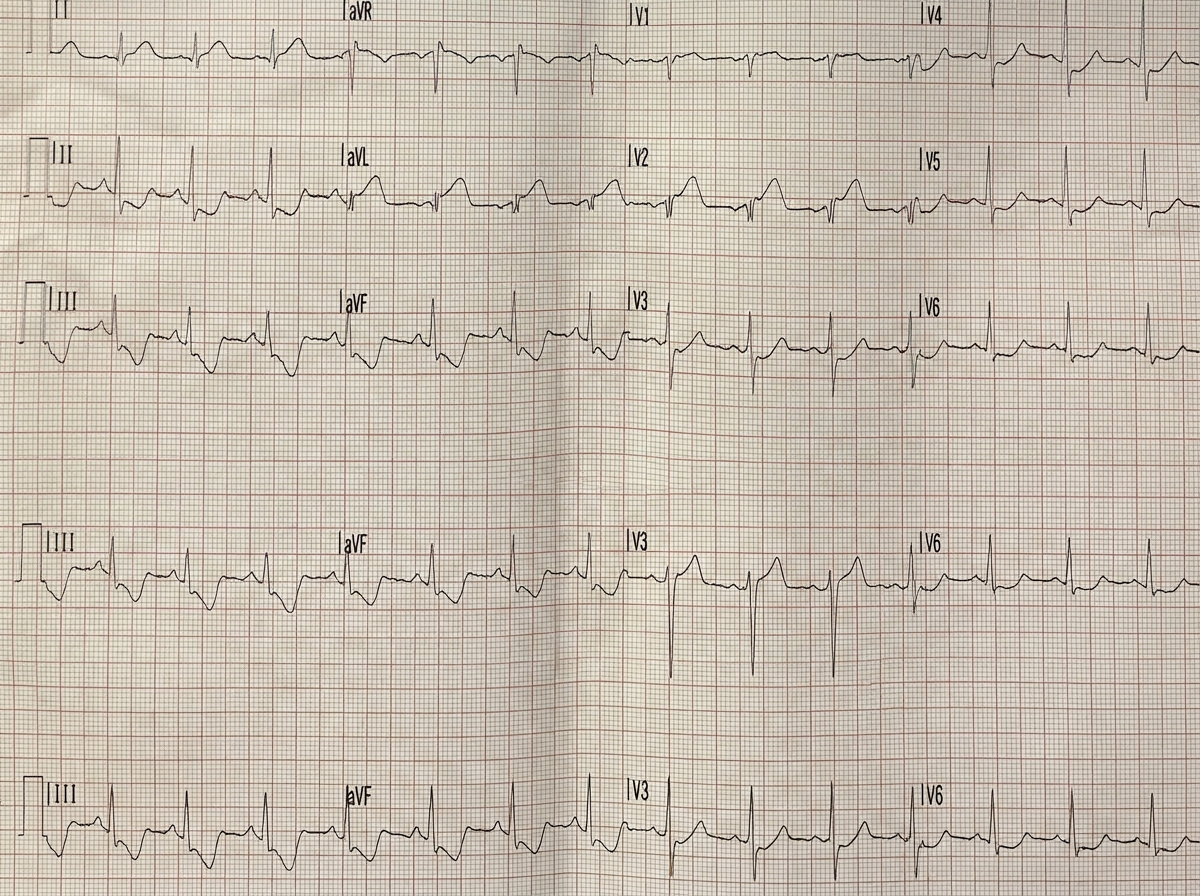
A 73-year-old woman arrives at the emergency department due to intense central chest pain for 30 minutes this morning. She says the pain was cramping in nature and radiated down her left arm. She has a history of atrial fibrillation and type 2 diabetes mellitus. Her pulse is 98/min, respiratory rate is 19/min, temperature is 36.8°C (98.2°F), and blood pressure is 160/91 mm Hg. Cardiovascular examination shows no abnormalities. ECG is shown below. Which of the following biochemical markers would most likely be elevated and remain elevated for a week after this acute event?
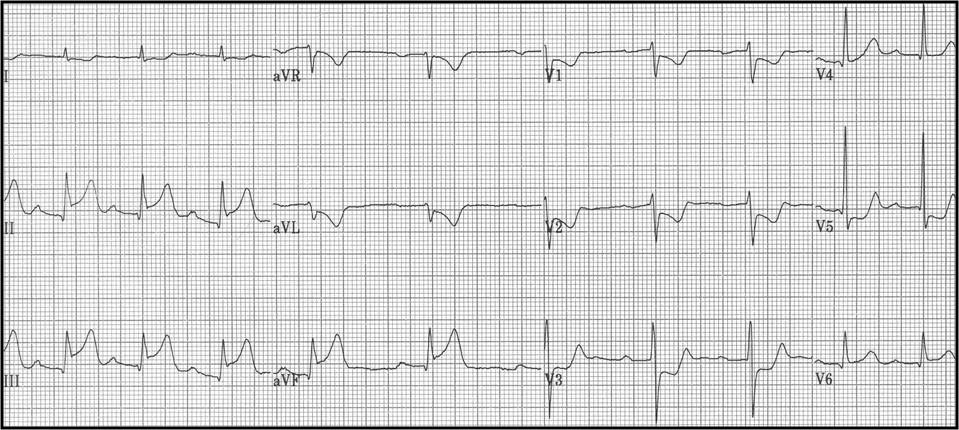
A 55-year-old woman is brought to the emergency department by her husband because of chest pain and a cough productive of blood-tinged sputum that started 1 hour ago. Two days ago, she returned from a trip to China. She has smoked 1 pack of cigarettes daily for 35 years. Her only home medication is oral hormone replacement therapy for postmenopausal hot flashes. Her pulse is 123/min and blood pressure is 91/55 mm Hg. Physical examination shows distended neck veins. An ECG shows sinus tachycardia, a right bundle branch block, and T-wave inversion in leads V5–V6. Despite appropriate lifesaving measures, the patient dies. Examination of the lung on autopsy shows a large, acute thrombus in the right pulmonary artery. Based on the autopsy findings, which of the following is the most likely origin of the thrombus?
A 60-year-old African American gentleman presents to the emergency department with sudden onset "vice-like" chest pain, diaphoresis, and pain radiating to his left shoulder. He has ST elevations on his EKG and elevated cardiac enzymes. Concerning his current pathophysiology, which of the following changes would you expect to see in this patient?
A 56-year-old man comes to the emergency department because of chest pain. The pain occurs intermittently in 5-minute episodes. It is not conclusively brought on by exertion and sometimes occurs at rest. He has a history of hyperlipidemia and takes a high-dose statin daily. His father died of lung cancer at the age of 67 years and his mother has type 2 diabetes. He smokes a pack of cigarettes daily and does not drink alcohol. His temperature is 37°C (98.8°F), pulse is 88/min, and blood pressure is 124/72 mm Hg. Cardiac examination shows no abnormalities. He has no chest wall tenderness and pain is not reproduced with palpation. While waiting for laboratory results, he has another episode of chest pain. During this event, an ECG shows ST elevations in leads II, III, and aVF that are > 1 mm. Thirty minutes later, a new ECG shows no abnormalities. Troponin I level is 0.008 ng/mL (normal value < 0.01 ng/mL). Cardiac angiography is performed and shows a 30% blockage of the proximal right circumflex artery and 10% blockage in the distal left circumflex artery. This patient's condition is most closely associated with which of the following?
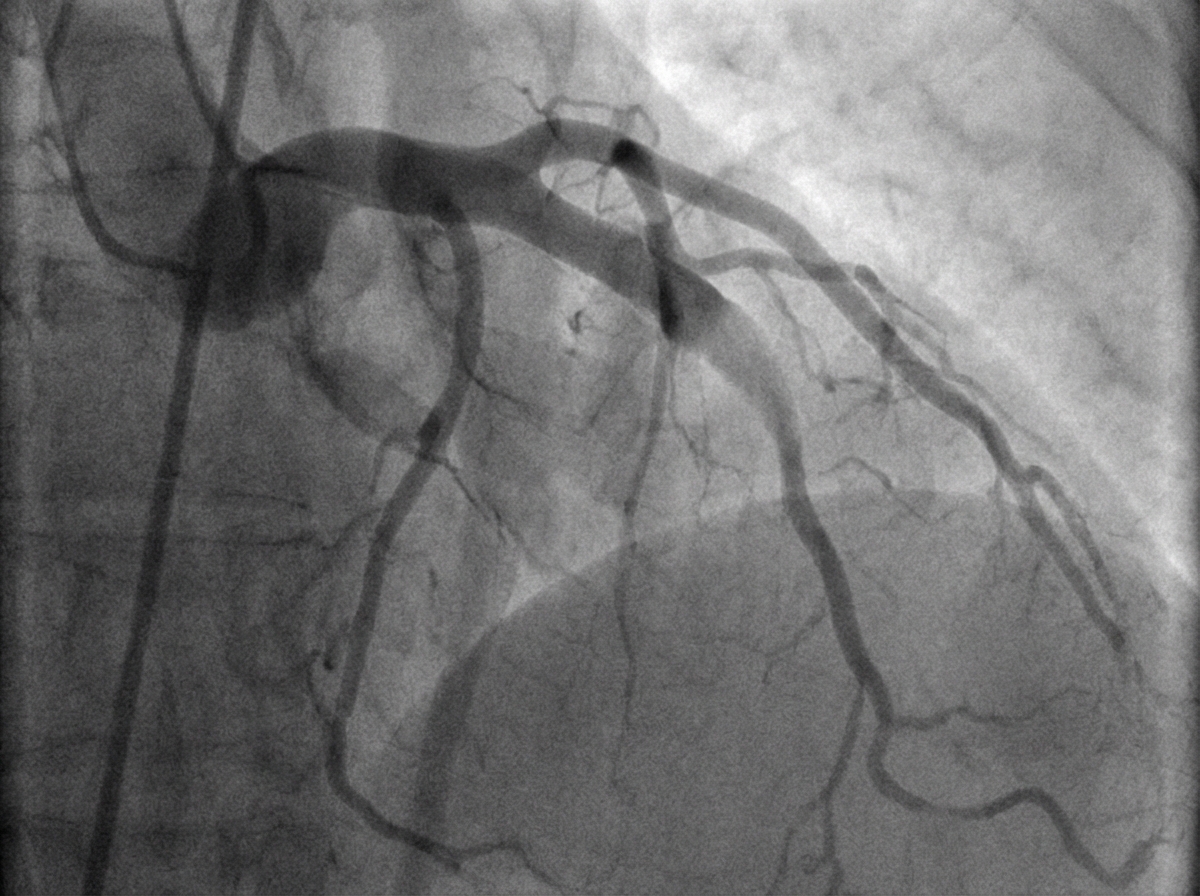
A 54-year-old woman is brought to the emergency department because of a 2-hour history of nausea and retrosternal chest pain. She has a 15-year history of type 2 diabetes mellitus. Her current medications include atorvastatin, metformin, and lisinopril. She is diaphoretic. Her serum troponin level is 3.0 ng/mL (N: < 0.04). She undergoes cardiac catheterization. A photograph of coronary angiography performed prior to percutaneous coronary intervention is shown. An acute infarct associated with the finding on angiography is most likely to manifest with ST elevations in which of the following leads on ECG?
Two days after coronary artery stent placement for a posterior myocardial infarction, a 70-year-old woman complains of difficulty breathing and retrosternal chest pain. She has a history of atrial fibrillation, for which she takes verapamil. Following stent placement, the patient was started on aspirin and clopidogrel. She appears to be in acute distress and is disoriented. Respirations are 22/min. Pulse oximetry on room air shows an oxygen saturation of 80%. Diffuse crackles are heard on auscultation of the chest. The patient is intubated and mechanical ventilation is started. Shortly afterwards, she becomes unresponsive. Heart sounds are inaudible and her carotid pulses are not palpable. The cardiac monitor shows normal sinus rhythm with T-wave inversion. Which of the following is the most appropriate next step in management?
A 57-year-old man presents to his primary care provider because of chest pain for the past 3 weeks. The chest pain occurs after climbing more than 2 flights of stairs or walking for more than 10 minutes. His symptoms remain for an average of 30 minutes despite rest, but they eventually remit. He is obese, has a history of type 2 diabetes mellitus, and has smoked 15–20 cigarettes a day for the past 25 years. His father died from a myocardial infarction at 52 years of age. His vital signs reveal a temperature of 36.7°C (98.0°F), blood pressure of 145/93 mm Hg, and a heart rate of 85/min. The physical examination is unremarkable. Which of the following is consistent with unstable angina?
Practice by Chapter
ACS pathophysiology and classification
Practice Questions
STEMI diagnosis and management
Practice Questions
NSTEMI diagnosis and management
Practice Questions
Unstable angina
Practice Questions
Cardiac biomarkers
Practice Questions
ECG interpretation in ACS
Practice Questions
Reperfusion strategies
Practice Questions
Antiplatelet therapy
Practice Questions
Anticoagulation in ACS
Practice Questions
Complications of MI
Practice Questions
Secondary prevention
Practice Questions
Special populations (elderly, renal dysfunction)
Practice Questions
Risk stratification in ACS
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
