Cardiovascular System — MCQs

On this page
Which part of ventricular action potential corresponds to ST segment in ECG?
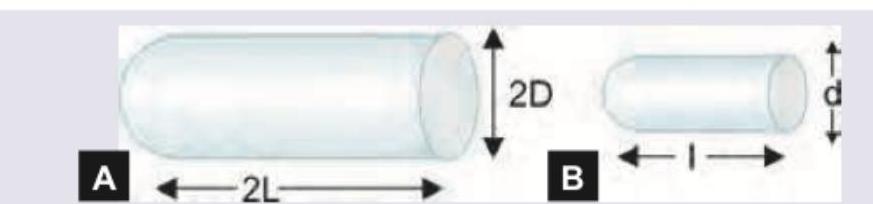
There are two blood vessels shown below. Assuming that pressure along both the vessels is same and both of them follow linear flow pattern, what will be the amount of blood flow in A compared to B?
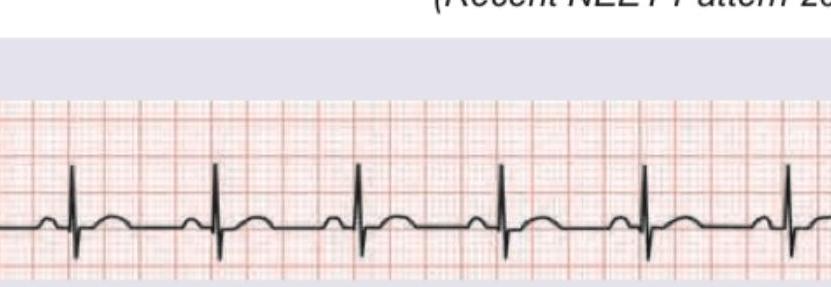
Compare the two ECG recordings taken before and after activation of low pressure atrial stretch receptors. Which reflex explains the findings?
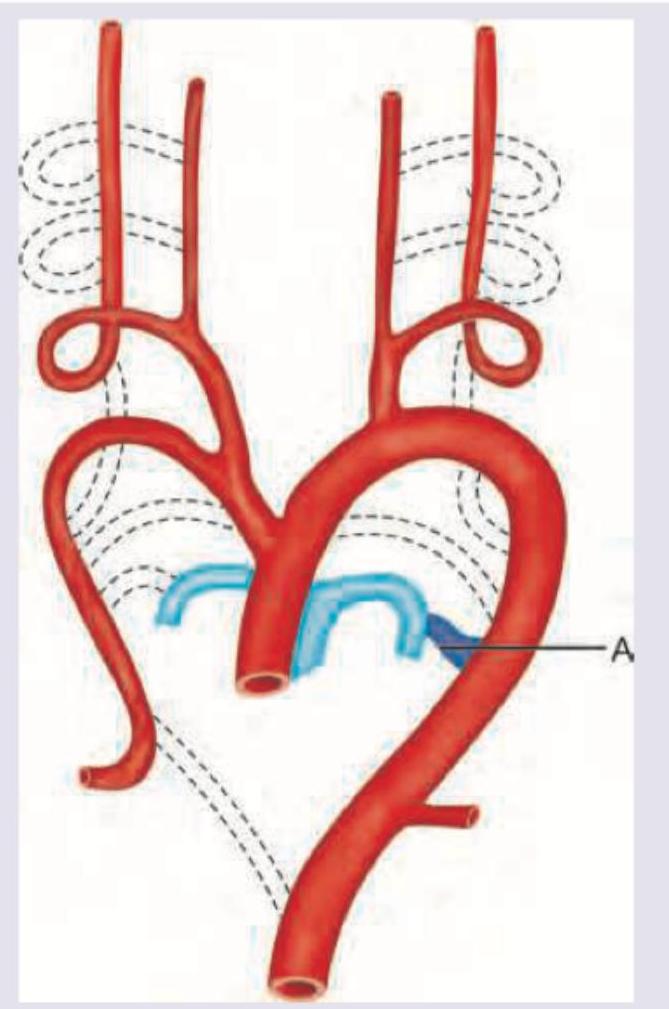
The structure marked $A$ begins to close by what time frame and due to what cause?
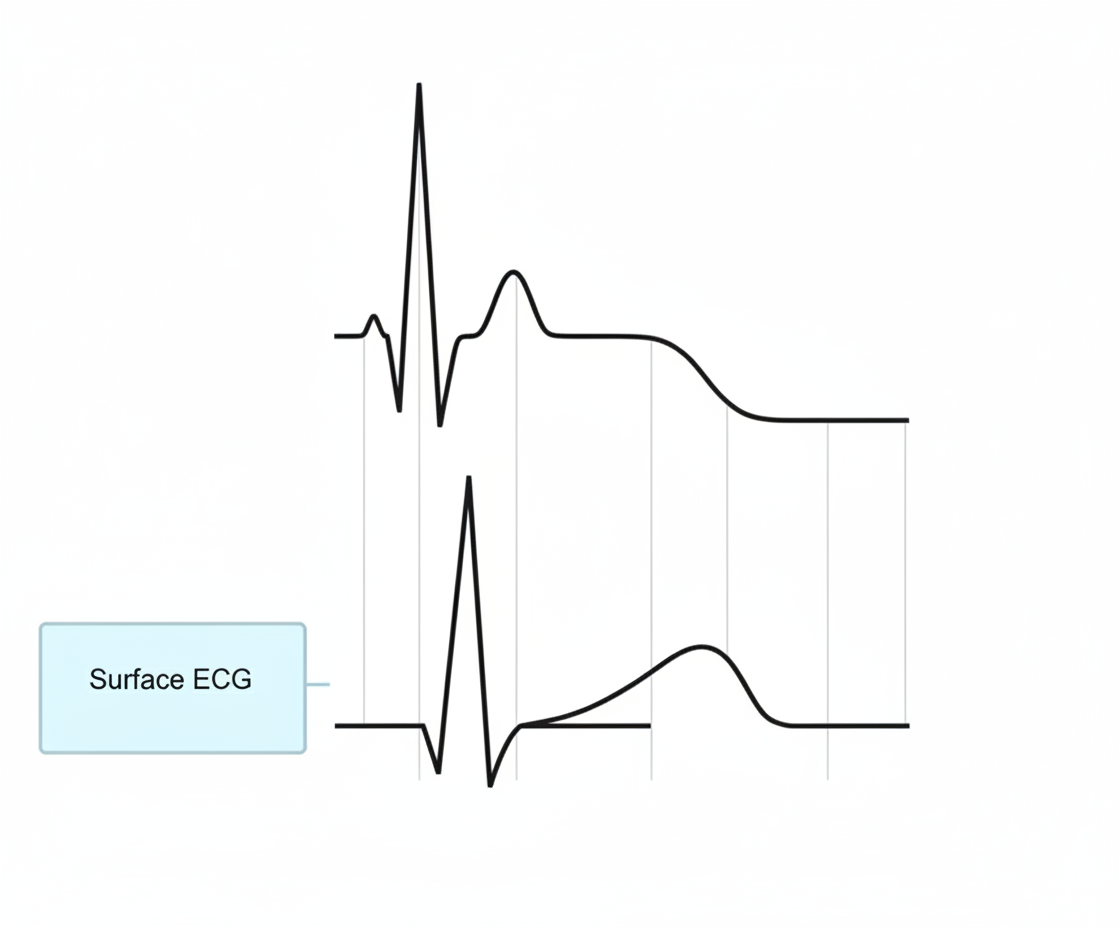
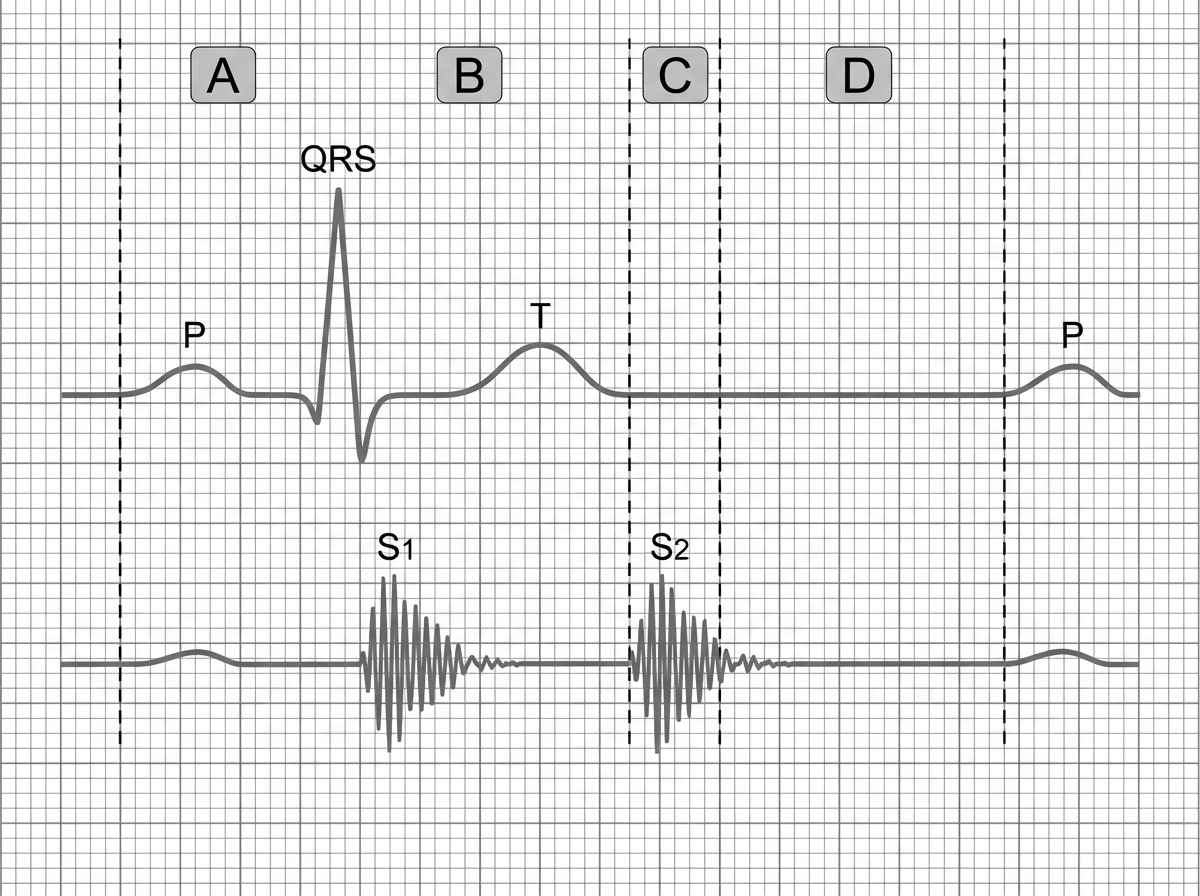
Phonocardiogram tracing is shown below with corresponding ECG. Identify the phase corresponding with $S_{2}$ in phonocardiogram.
Using the quadrant method, if the mean QRS vector in lead I is negative and in lead aVF is positive, what is the axis?
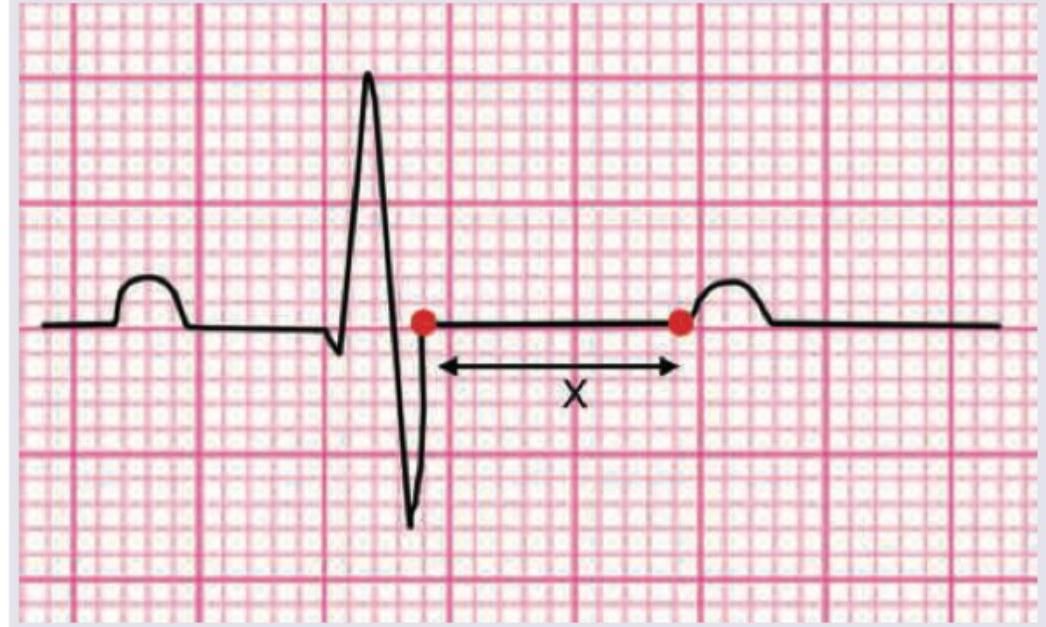
The marked part of the ECG called as 'X' points to which phase of cardiac action potential?
Smoking is considered to be a modifiable risk factor for Coronary Heart Disease. Consider the following statements with regard to possible mechanisms on the basis of which it acts as a risk factor : 1. Nicotine stimulation of adrenergic drive raises the blood pressure and myocardial oxygen demand. 2. It increases carbon monoxide and induces atherogenesis. 3. It leads to fall in protective high density lipoproteins. 4. It reduces the apolipoprotein-B plasma levels. Which of the statements given above are correct ?
Distributive shock is described by which of the following patterns of cardiovascular responses? 1. Vasodilation 2. Reduced peripheral vascular resistance 3. Inadequate 'afterload' 4. Low cardiac output Select the correct answer using the code given below.
Non-cardiac causes of raised central venous pressure include all of the following except:
Practice by Chapter
Cardiac Electrophysiology
Practice Questions
Cardiac Cycle
Practice Questions
Cardiac Output and Its Regulation
Practice Questions
Hemodynamics and Blood Flow
Practice Questions
Arterial System Physiology
Practice Questions
Microcirculation and Lymphatics
Practice Questions
Venous Return and Central Venous Pressure
Practice Questions
Cardiovascular Reflexes
Practice Questions
Regional Circulations
Practice Questions
Cardiovascular Responses to Exercise and Stress
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
