Pharmacokinetics and Pharmacodynamics — MCQs

On this page
Which of the following vaccines are given through the intramuscular route? 1. BCG vaccine 2. Hepatitis B vaccine 3. Pentavalent vaccine 4. Inactivated Polio vaccine
Which one of the following statements regarding the composition of common crystalloid solutions is correct ?
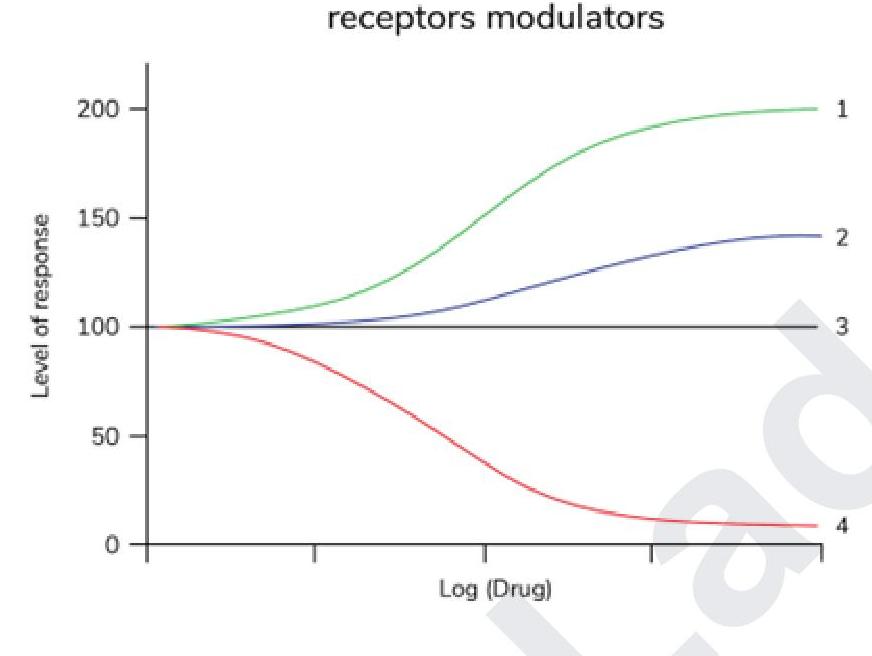
Which of the following statements is correct regarding the given graph?
Choose the correct options regarding the route of administration and bioavailability. A- Intravenous =1 B- 0.75< Oral <1 C-0.75 <IM ≤ 1 D- 0.75<SC ≤ 1 IM - Intramuscular SC- Subcutaneous
What would happen to the half-life and plasma concentration of a drug which follows first-order kinetics, if the dose is doubled?
Purpose of adding zinc to insulin in commercial preparations for treatment of diabetes mellitus is:-
True for lithium:
The shortest acting opioid is:-
Atorvastatin is used as an anti-dyslipidemic drug. These drugs inhibit their target enzyme by:-
Therapeutic index of a drug is an indicator of:-
Practice by Chapter
Absorption and Bioavailability
Practice Questions
Drug Distribution and Protein Binding
Practice Questions
Biotransformation and Metabolism Pathways
Practice Questions
Renal and Non-renal Excretion
Practice Questions
Compartment Models
Practice Questions
Dose-Response Relationships
Practice Questions
Drug Efficacy and Potency
Practice Questions
Drug Tolerance and Tachyphylaxis
Practice Questions
Population Pharmacokinetics
Practice Questions
Pharmacokinetic Variability
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
