Pharmacokinetics and Pharmacodynamics — MCQs

On this page
Which of the following does NOT induce microsomal enzymes?
Which of the following cephalosporins does not require dose modifications even in the presence of decreased GFR?
When the same dose of a drug is repeated at half-life intervals, after how many half-lives is the steady-state (plateau) plasma drug concentration reached?
When atrial fibrillation persists despite digoxin therapy, an inadequate digoxin dose is suspected. Plasma levels of digoxin for confirmation are typically drawn after a certain interval following the last dose. Considering pharmacokinetic principles, which factor is most crucial for determining this appropriate sampling time?
Single dose aminoglycoside administration is more preferable than 8 hourly dosing because of which of the following?
Which of the following is false regarding spare receptors?
Which of the following is NOT a prodrug?
Which of the following is a prodrug of cetirizine?
Which of the following is a CYP-450 inducer?
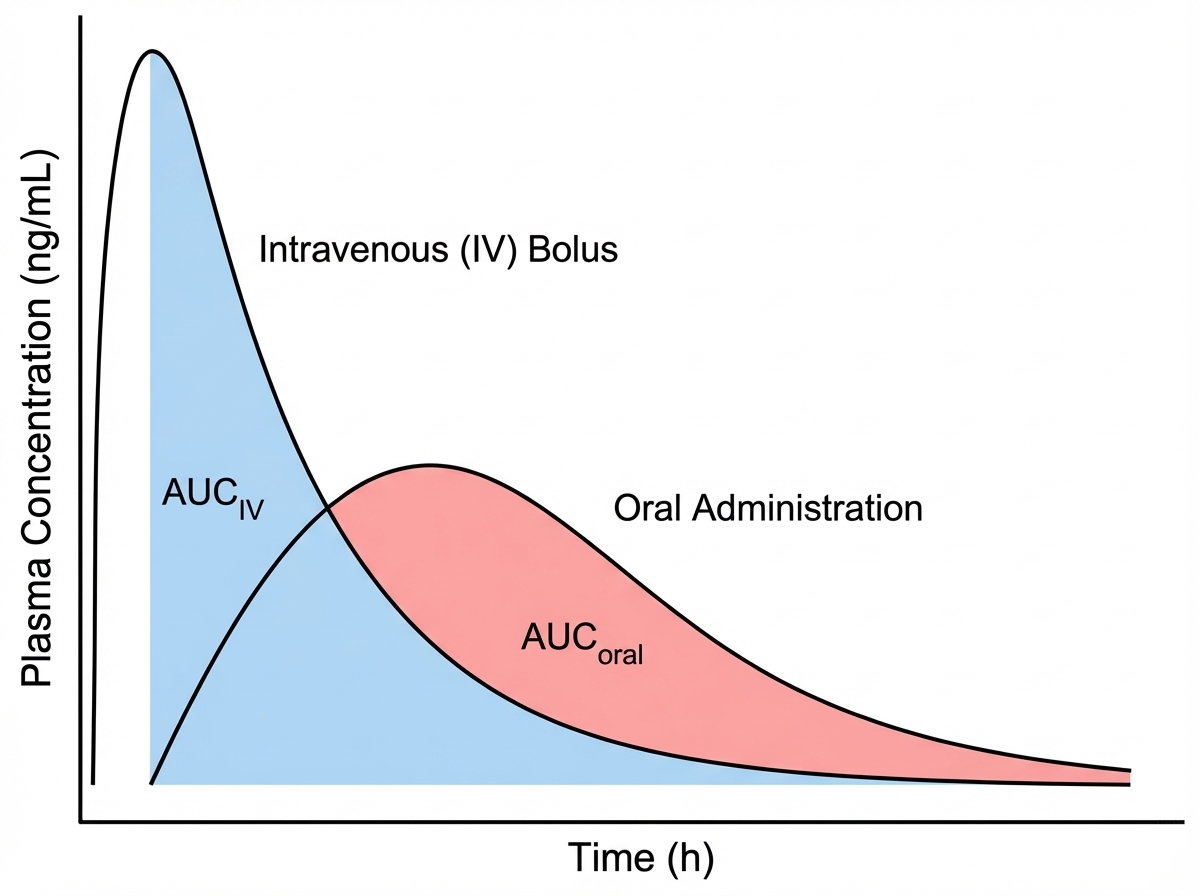
What does the plasma concentration versus time graph indicate?
Practice by Chapter
Absorption and Bioavailability
Practice Questions
Drug Distribution and Protein Binding
Practice Questions
Biotransformation and Metabolism Pathways
Practice Questions
Renal and Non-renal Excretion
Practice Questions
Compartment Models
Practice Questions
Dose-Response Relationships
Practice Questions
Drug Efficacy and Potency
Practice Questions
Drug Tolerance and Tachyphylaxis
Practice Questions
Population Pharmacokinetics
Practice Questions
Pharmacokinetic Variability
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
