Neoplasia — MCQs

On this page
In a patient with non-Hodgkin B cell lymphoma, a nuclear gene is actively transcribed to mRNA and transported to the cytoplasm, where a protein is translated. This process is associated with up-regulation of BCL2. In a control group without lymphoma, translation of this mRNA does not occur. How is the silencing of this active gene's mRNA most likely to occur?
Which type of cancer is most commonly associated with radiation exposure?
Histopathological examination reveals the diagnosis as:
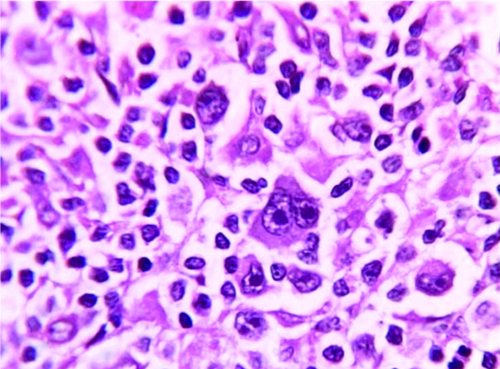
Which type of breast carcinoma is characterized by a pushing border?
In Wilms tumor, which of the following leads to emergence of resistance to chemotherapy?
Endodermal sinus tumor is associated with which of the following?
Which of the following immunohistochemical markers is positive in neuroendocrine tumors?
S100 is a marker of which of the following?
What is the most common extragonadal site for germ cell tumors?
A patient complains of epigastric pain that fails to respond to antacids. Endoscopy demonstrates an ulcerated mass on the greater curvature of the stomach. Genetic studies on the tumor demonstrate an altered DCC gene. Which of the following tumor suppressor genes is found on the same chromosome as DCC?
Practice by Chapter
Nomenclature and Classification of Tumors
Practice Questions
Characteristics of Benign and Malignant Neoplasms
Practice Questions
Molecular Basis of Cancer
Practice Questions
Carcinogenesis and Carcinogens
Practice Questions
Tumor Progression and Metastasis
Practice Questions
Tumor Markers
Practice Questions
Paraneoplastic Syndromes
Practice Questions
Genetic Basis of Cancer
Practice Questions
Tumor Immunity
Practice Questions
Cancer Epidemiology and Prevention
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
