Pulmonology — MCQs

On this page
Which of the following is NOT a complication of bronchiectasis?
Which of the following diagnostic techniques is most specific for pulmonary embolism?
Hemoptysis may occur in which of the following situations except?
A 65-year-old man presents with a chronic cough, which has recently changed character. Tuberculosis and other infectious causes have been ruled out. What should be the next investigation?
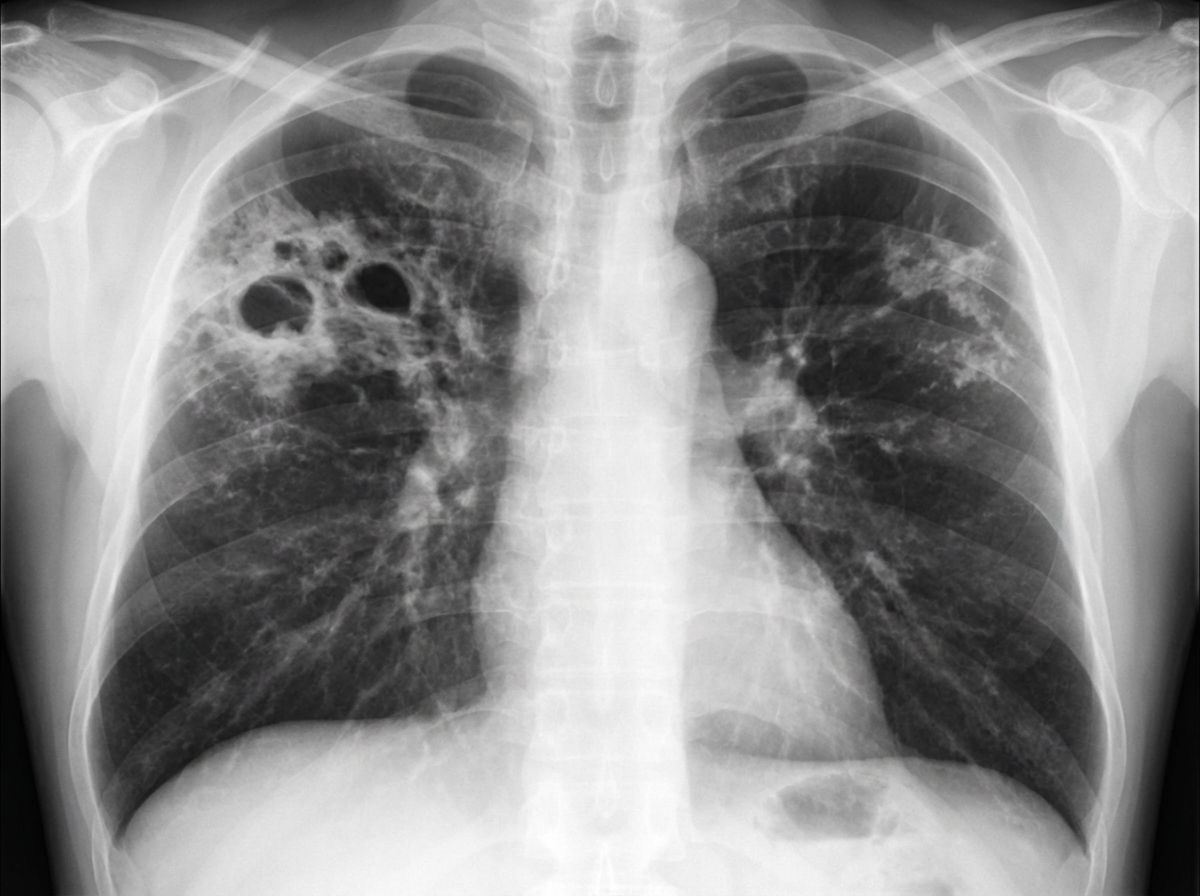
A 60-year-old man with a history of COPD and old TB presents with mild hemoptysis and chronic cough. He is HIV negative and has been ill for approximately 2 weeks. Vital signs: pulse 110 bpm; temperature 101°F; respirations 24/min; blood pressure 108/70 mm Hg. No skin lesions are noted. Laboratory data: Hb 14 g/dL; HCT 42%; WBCs 8.7/uL; BUN 24 mg/dL; creatinine 0.8 mg/dL; sodium 131 mEq/L; potassium 4.3 mEq/L. Arterial blood gases on room air: pH 7.37; PCO2 43 mm Hg; PO2 87 mm Hg. Sputum tests reveal numerous AFB-positive organisms on smear. Spirometry shows an obstructive ventilatory impairment with marginal reversibility. Chest X-ray is provided. Among the choices listed, what is the most likely diagnosis?
Which of the following interstitial lung diseases is not associated with smoking?
Which one of the following is not likely to be associated with pulmonary fibrosis?
Central cyanosis is seen if?
A 32-year-old patient presents with complaints of difficulty in breathing, chest pain, and increased respiratory rate. Pulmonary thromboembolism is suspected. Which investigation is most useful in acute pulmonary thromboembolism?
All of the following are features of alveolitis (interstitial lung disease), except?
Practice by Chapter
Obstructive Airway Diseases (Asthma, COPD)
Practice Questions
Interstitial Lung Diseases
Practice Questions
Pulmonary Infections
Practice Questions
Pulmonary Vascular Diseases
Practice Questions
Pleural Diseases
Practice Questions
Sleep-Disordered Breathing
Practice Questions
Respiratory Failure
Practice Questions
Mediastinal Disorders
Practice Questions
Occupational Lung Diseases
Practice Questions
Pulmonary Function Testing
Practice Questions
Bronchiectasis and Cystic Fibrosis
Practice Questions
Lung Cancer Approach
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
