Nephrology — MCQs

On this page
Which of the following biomarkers is not typically involved in the diagnosis of acute kidney injury?
True about Bartter's syndrome are all except?
Which of the following primary kidney diseases is NOT typically associated with nephrotic syndrome?
Which of the following is most commonly affected in Ormond's disease?
Most common cause of hypernatremia.
Chronic hemodialysis in ESRD patient is done
A 40-year-old man with a known case of hypertension presented with multiple episodes of hematuria and loin pain. His elder brother passed away due to a stroke at the age of 40. What is the probable diagnosis based on the clinical presentation?
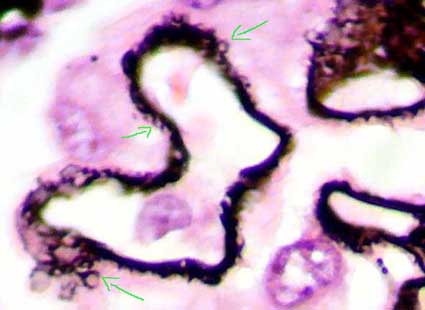
A 48-year-old man presents with complaints of facial puffiness, frothy urine, and hypertension. He gives a history of infection with hepatitis B. Urine examination reveals microscopic hematuria. The histopathological image of the kidney biopsy shows a spike and dome pattern. What is the diagnosis of this condition?
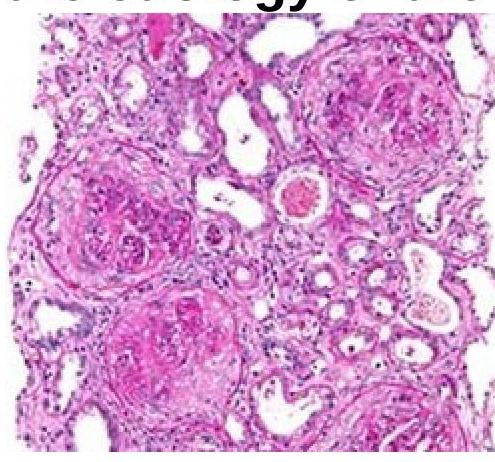
A 51-year-old person came with a complaint of hematuria. On examination, he was normotensive and had pedal edema. Investigations revealed the patient had no glucosuria and had a creatinine value of 9mg%. Renal biopsy is as shown below. Which of the following investigations should be done to identify the etiology of the disease?
What is the most likely cause of metabolic acidosis in a patient with a pH of 7.2, HCO3- levels of 10 or 12, and pCO2 of 35 mmHg?
Practice by Chapter
Acute Kidney Injury
Practice Questions
Chronic Kidney Disease
Practice Questions
Glomerular Diseases
Practice Questions
Tubulointerstitial Diseases
Practice Questions
Nephrotic and Nephritic Syndromes
Practice Questions
Urinary Tract Infections
Practice Questions
Renal Replacement Therapy
Practice Questions
Fluid and Electrolyte Disorders
Practice Questions
Acid-Base Disorders
Practice Questions
Kidney in Systemic Diseases
Practice Questions
Kidney Stones and Obstructive Uropathy
Practice Questions
Hypertension in Kidney Disease
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
