Nephrology — MCQs

On this page
A 47-year-old HIV-positive man presents with weakness, HIV nephropathy, and adrenal insufficiency. He is taking trimethoprim-sulfamethoxazole for PCP prophylaxis and triple-agent antiretroviral treatment. He was recently started on spironolactone for ascites due to alcoholic liver disease. Physical examination reveals normal vital signs, but his muscles are diffusely weak. Frequent extrasystoles are noted. He has mild ascites and 1+ peripheral edema. Laboratory studies show a serum creatinine of 2.5 mg/dL with a potassium value of 7.3 mEq/L. ECG shows peaking of the T-waves and QRS widening to 0.14 seconds. Once the patient is stabilized and the T-waves have normalized, it is important to review the potential causes of his hyperkalemia and to take steps to prevent this from happening again. As you consider the pathophysiology of each factor, which of the following statements is true?
A patient being treated for leukemia develops unilateral flank pain. Radiologic studies demonstrate a dilated renal pelvis and dilation of the upper one-third of the corresponding ureter. A stone with which of the following compositions is most likely causing this patient's problems?
All of the following are true regarding Bartter syndrome except?
All of the following are true about Bartter syndrome except?
A 57-year-old male is admitted to the hospital for a suspected kidney infection (likely pyelonephritis). The patient is placed on intravenous antibiotic therapy but continues to have a temperature of 103°F after 3 days of therapy. The urine culture grows lactose-fermenting Gram-negative bacilli which are pan drug-sensitive. On examination, he appears ill and has marked left flank tenderness. Ultrasound depicts a specific finding. What is the most likely diagnosis?
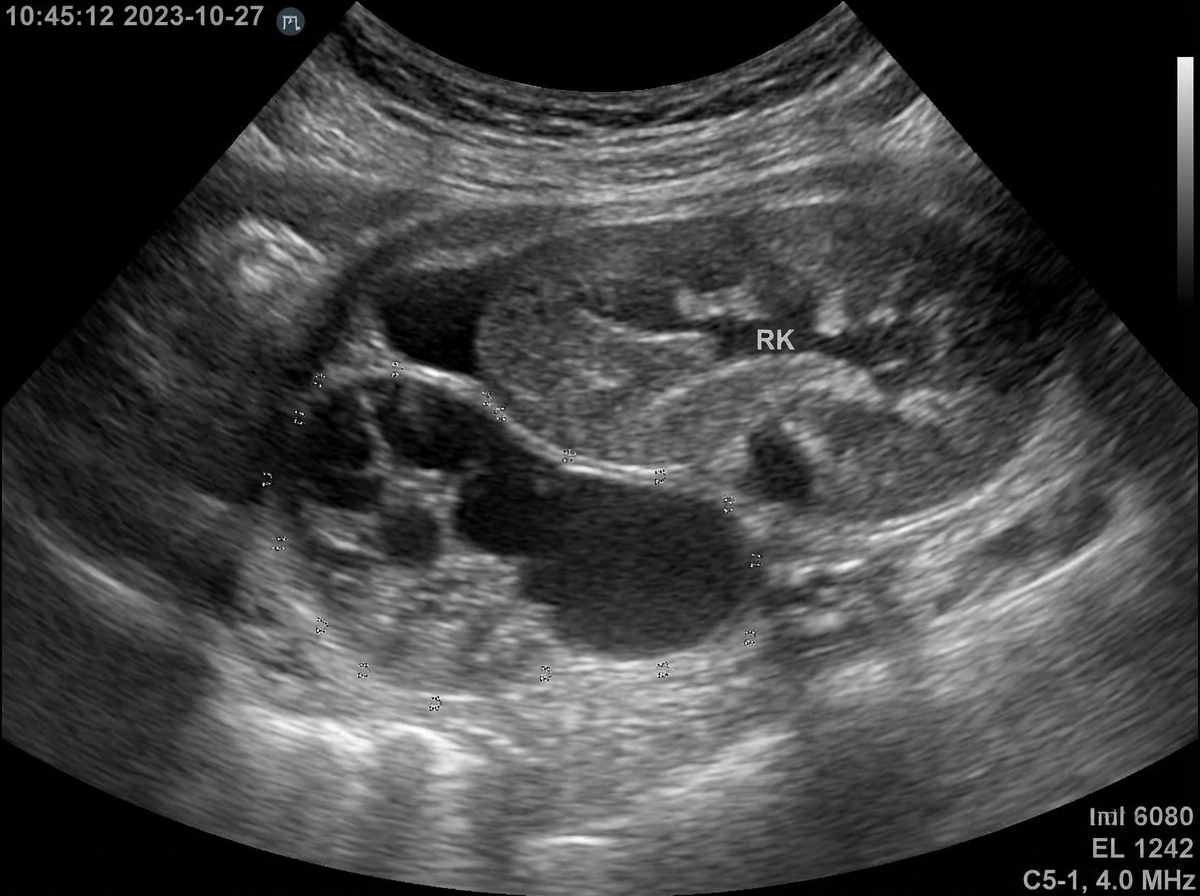
A 24-year-old man has a urine analysis that is positive for blood. The microscopic examination of the urine is negative for casts or red blood cells. Which of the following is the most likely diagnosis?
What is the most common cause of renal papillary necrosis?
Normal anion gap metabolic acidosis can occur in which of the following conditions?
Which of the following statements about kidney injury molecule-1 (KIM-1) is false?
Which of the following is seen in nephrotic syndrome?
Practice by Chapter
Acute Kidney Injury
Practice Questions
Chronic Kidney Disease
Practice Questions
Glomerular Diseases
Practice Questions
Tubulointerstitial Diseases
Practice Questions
Nephrotic and Nephritic Syndromes
Practice Questions
Urinary Tract Infections
Practice Questions
Renal Replacement Therapy
Practice Questions
Fluid and Electrolyte Disorders
Practice Questions
Acid-Base Disorders
Practice Questions
Kidney in Systemic Diseases
Practice Questions
Kidney Stones and Obstructive Uropathy
Practice Questions
Hypertension in Kidney Disease
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
