Nephrology — MCQs

On this page
All of the following are associated with low complement levels, EXCEPT:
Which of the following tests is most sensitive for detecting early diabetic nephropathy?
What is the cause of hypercoagulation in nephrotic syndrome?
Which is the most common electrolyte abnormality causing seizures in hospitalized patients?
A 64-year-old man developed persistent back pain. On examination, his BP is 120/80, pulse 70, respiratory rate is 15, and temp is 98.6 deg F. Physical findings include severe pallor and evidence of muscle wasting. Urinalysis shows 4+ protein, and microscopy reveals occasional broad and occasional granular casts. Lab studies include: Serum creatinine = 5.1 mg/dl, serum Na = 141 mEq/L, K = 5.6 mEq/L, chloride = 101 mEq/L, CO2 = 14 mEq/L, serum calcium = 11.7 mg/dl, and serum phosphorus = 6.0 mg/dl. What is the most likely etiology?
What is the most rapid method for lowering serum potassium levels?
Which of the following drugs are implicated in the causation of nephrotic syndrome?
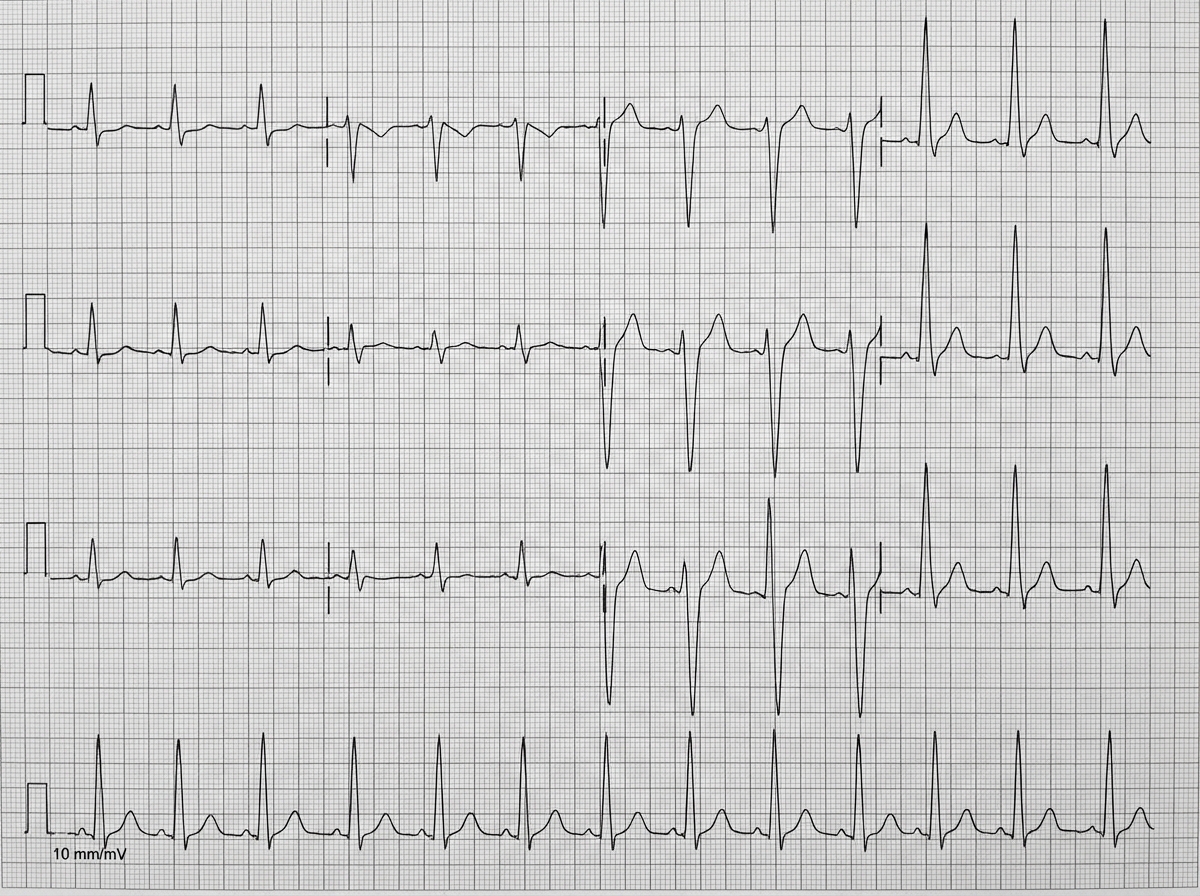
A 78-year-old man with chronic kidney disease presents with generalized malaise. His past medical history includes heart failure, hypertension, and type 2 diabetes. Medications are ramipril, insulin, furosemide, and metoprolol. On physical examination, the blood pressure is 155/90 mm Hg, heart rate is 100/min, and respiration 24/min. The heart sounds are normal, there is no edema, and the lungs are clear on auscultation. An ECG is performed. What is the most likely diagnosis based on the ECG findings?
Plasmapheresis is indicated in each of the following conditions except?
Azotemia occurs when:
Practice by Chapter
Acute Kidney Injury
Practice Questions
Chronic Kidney Disease
Practice Questions
Glomerular Diseases
Practice Questions
Tubulointerstitial Diseases
Practice Questions
Nephrotic and Nephritic Syndromes
Practice Questions
Urinary Tract Infections
Practice Questions
Renal Replacement Therapy
Practice Questions
Fluid and Electrolyte Disorders
Practice Questions
Acid-Base Disorders
Practice Questions
Kidney in Systemic Diseases
Practice Questions
Kidney Stones and Obstructive Uropathy
Practice Questions
Hypertension in Kidney Disease
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
