Nephrology — MCQs

On this page
Which of the following is not a feature of acute renal failure?
Adult polycystic kidney disease is inherited:
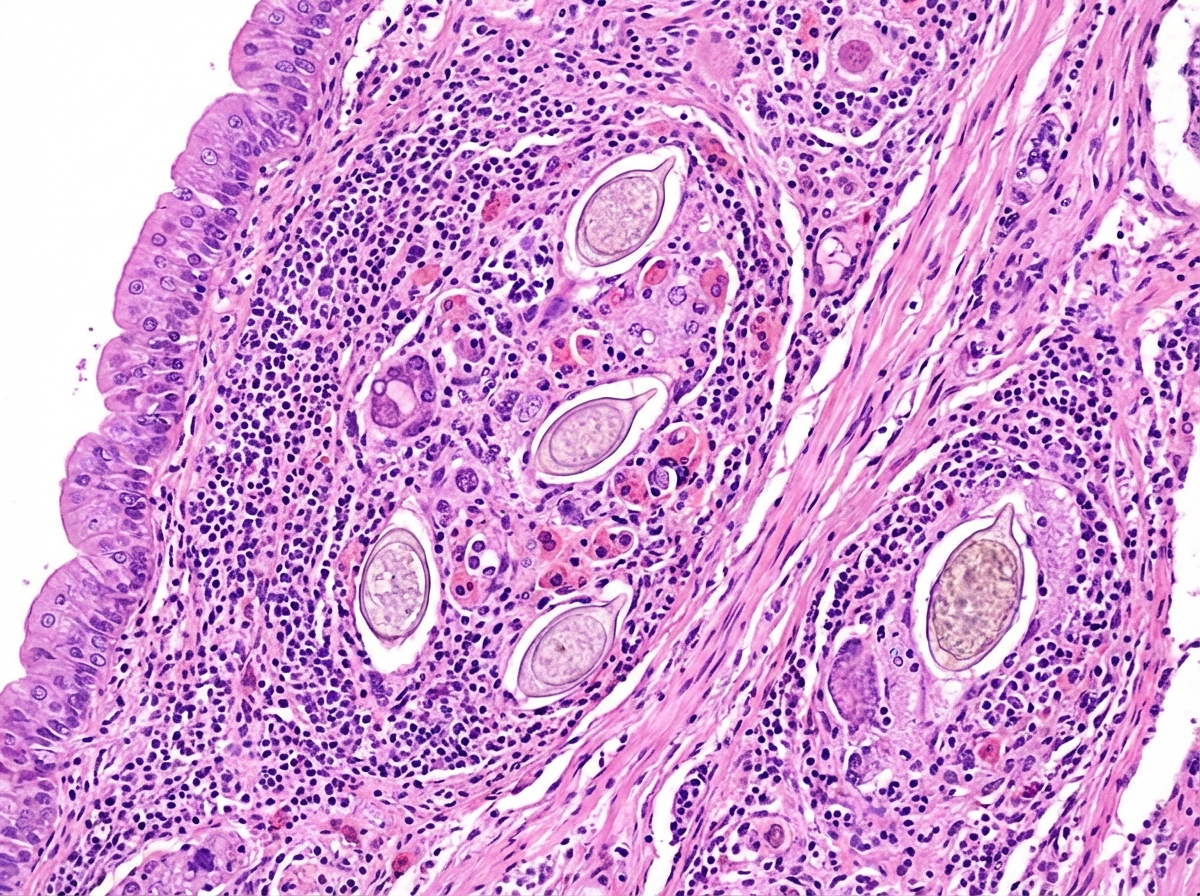
A 35-year-old male presented with gross hematuria, with similar episodes occurring in the past 6 months. There was no history of fever, pain, or burning on urination, weight loss, or increased sense of urgency to void. There was no history of exposure to toxins either at work or during recreational activities. The patient underwent fiberoptic cystoscopy, and a mass was noted in the fundus of the bladder. Biopsy of the mass is shown in the figure. What is the drug of choice for the likely cause?
All of the following are used for the management of hyperkalemia except?
Clinical features of chronic renal failure/uremia appear when renal function is reduced to what extent?
All are true for acute nephritic syndrome EXCEPT:
Hyperkalemia is a feature of which of the following conditions?
All of the following are examples of tubulointerstitial disorders of the kidney except?
Rhabdomyolysis is characterized by:
Which of the following is true about sepsis-related acute kidney injury?
Practice by Chapter
Acute Kidney Injury
Practice Questions
Chronic Kidney Disease
Practice Questions
Glomerular Diseases
Practice Questions
Tubulointerstitial Diseases
Practice Questions
Nephrotic and Nephritic Syndromes
Practice Questions
Urinary Tract Infections
Practice Questions
Renal Replacement Therapy
Practice Questions
Fluid and Electrolyte Disorders
Practice Questions
Acid-Base Disorders
Practice Questions
Kidney in Systemic Diseases
Practice Questions
Kidney Stones and Obstructive Uropathy
Practice Questions
Hypertension in Kidney Disease
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
