Hematology — MCQs

On this page
A patient is a known case of thalassemia. Which of the following viruses would be responsible for attacking progenitor cells and causing aplastic anemia?
A patient presents with tingling sensation in both legs, polyuria, and weight loss. The bone marrow aspirate findings are shown in the image. What is the most likely diagnosis?
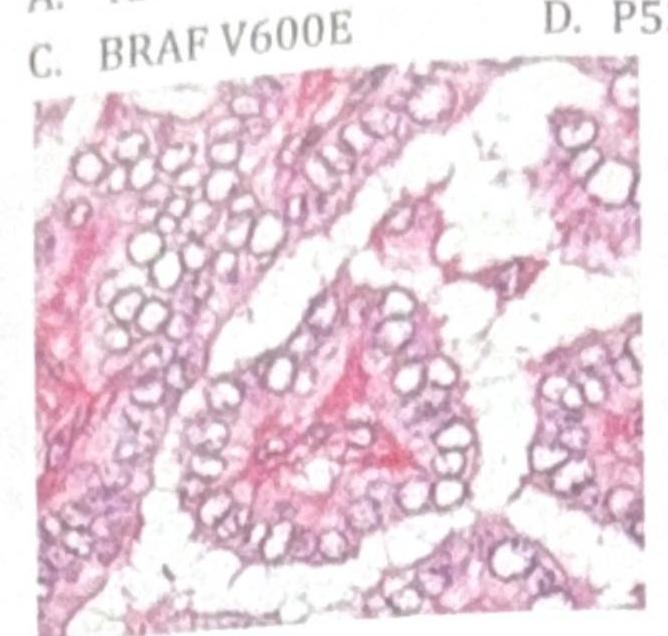
Which viral infection is most likely responsible for triggering this aplastic crisis in the patient?
Which of the following statements regarding von Willebrand disease is incorrect?
An 18-year-old male presents to the OPD with gum bleeding, fever, low total leukocyte count (TLC), and low platelet count. General examination is unremarkable. Further investigations reveal a low reticulocyte count, absent megakaryocytes, and no immature cells in the bone marrow. What is the most likely diagnosis?
Which of the following is considered a poor prognostic marker in multiple myeloma (MM)?
All are increased in IDA except
What is/are the characterstics of Iron defficiency Anemaia(IDA)?
Which of the following is not likely to be seen in a patient with Paroxysmal Nocturnal Hemoglobinuria (PNH)?
Which of the following can be prevented by transfusing irradiated RBCs?
Practice by Chapter
Anemia Evaluation and Management
Practice Questions
Hemoglobinopathies
Practice Questions
Thalassemias
Practice Questions
Platelet Disorders
Practice Questions
Coagulation Disorders
Practice Questions
Thrombotic Disorders
Practice Questions
Leukemias
Practice Questions
Lymphomas
Practice Questions
Multiple Myeloma and Plasma Cell Disorders
Practice Questions
Myeloproliferative Neoplasms
Practice Questions
Transfusion Medicine
Practice Questions
Hematopoietic Stem Cell Transplantation
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
