Hematology — MCQs

On this page
What is the other name for Cooley's anemia?
Which tumor is associated with polycythemia?
A 16-year-old female presents with generalized weakness and palpitations. Her Hb is 7 g/dL and peripheral smear shows microcytic hypochromic anemia. Reticulocyte count is 0.8%. Serum bilirubin is 1 mg%. What is the most likely diagnosis?
All of the following conditions cause thrombocytopenia except?
Secondary hemolytic anemia is seen in all except:
Which of the following statements about Schilling's test is false?
What is the most common cause of death in patients with Hemophilia B?
Hepatitis-associated aplastic anemia is caused by which type of hepatitis virus?
A 25-year-old female presented in December with chronic fatigue and cyanosis with bluish lips and arthralgia. Peripheral blood film is shown below. What is the likely cause?
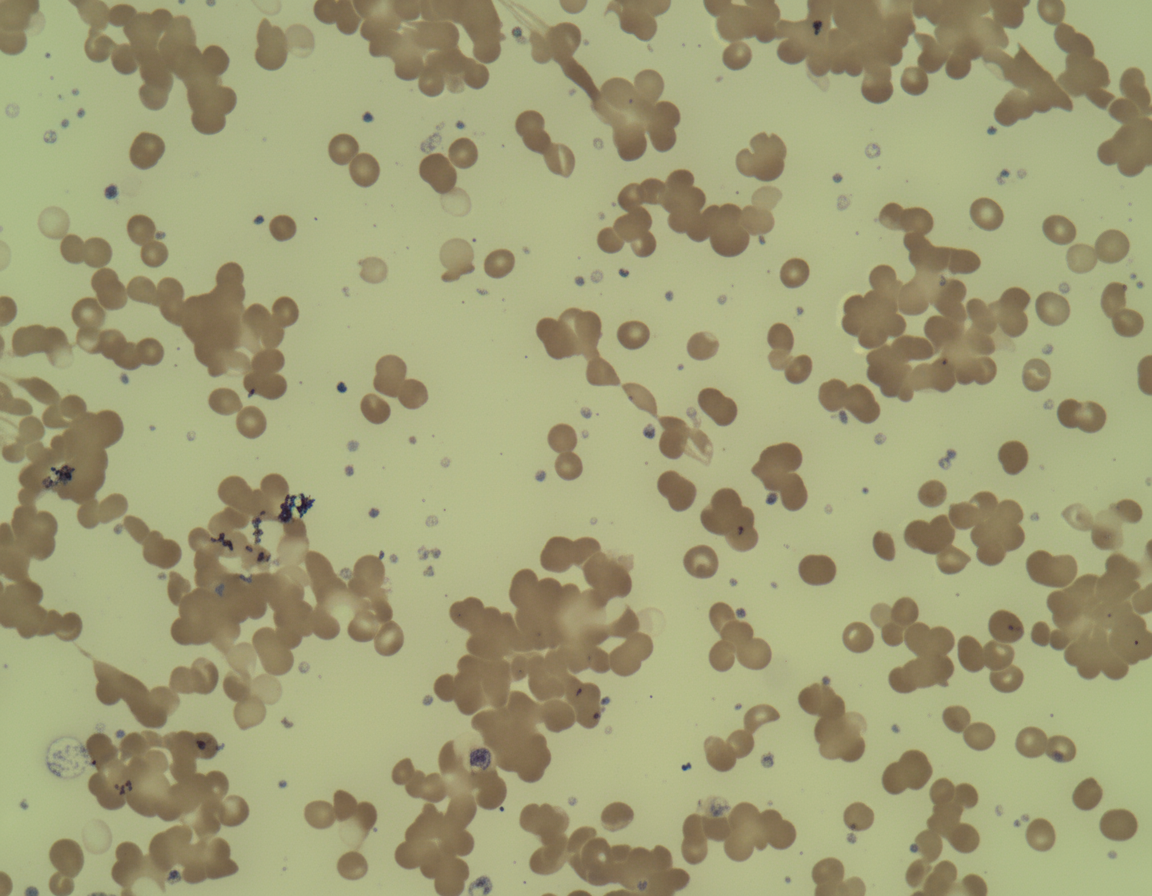
Upshaw-Schulman syndrome is classified as:
Practice by Chapter
Anemia Evaluation and Management
Practice Questions
Hemoglobinopathies
Practice Questions
Thalassemias
Practice Questions
Platelet Disorders
Practice Questions
Coagulation Disorders
Practice Questions
Thrombotic Disorders
Practice Questions
Leukemias
Practice Questions
Lymphomas
Practice Questions
Multiple Myeloma and Plasma Cell Disorders
Practice Questions
Myeloproliferative Neoplasms
Practice Questions
Transfusion Medicine
Practice Questions
Hematopoietic Stem Cell Transplantation
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
