Hematology — MCQs

On this page
Which of the following is NOT a feature of Antiphospholipid Syndrome (APS)?
The BCRABL gene mutation is characteristically associated with which of the following hematologic malignancies?
Which of the following sets of findings is seen in Disseminated Intravascular Coagulation (DIC)?
Which of the following is not associated with a high reticulocyte count?
A 69-year-old woman presents with fatigue. Her hemoglobin is 9.0 g/dL (14 g/dL one year prior) with no other symptoms. A blood film is provided. What is the most appropriate next diagnostic step?
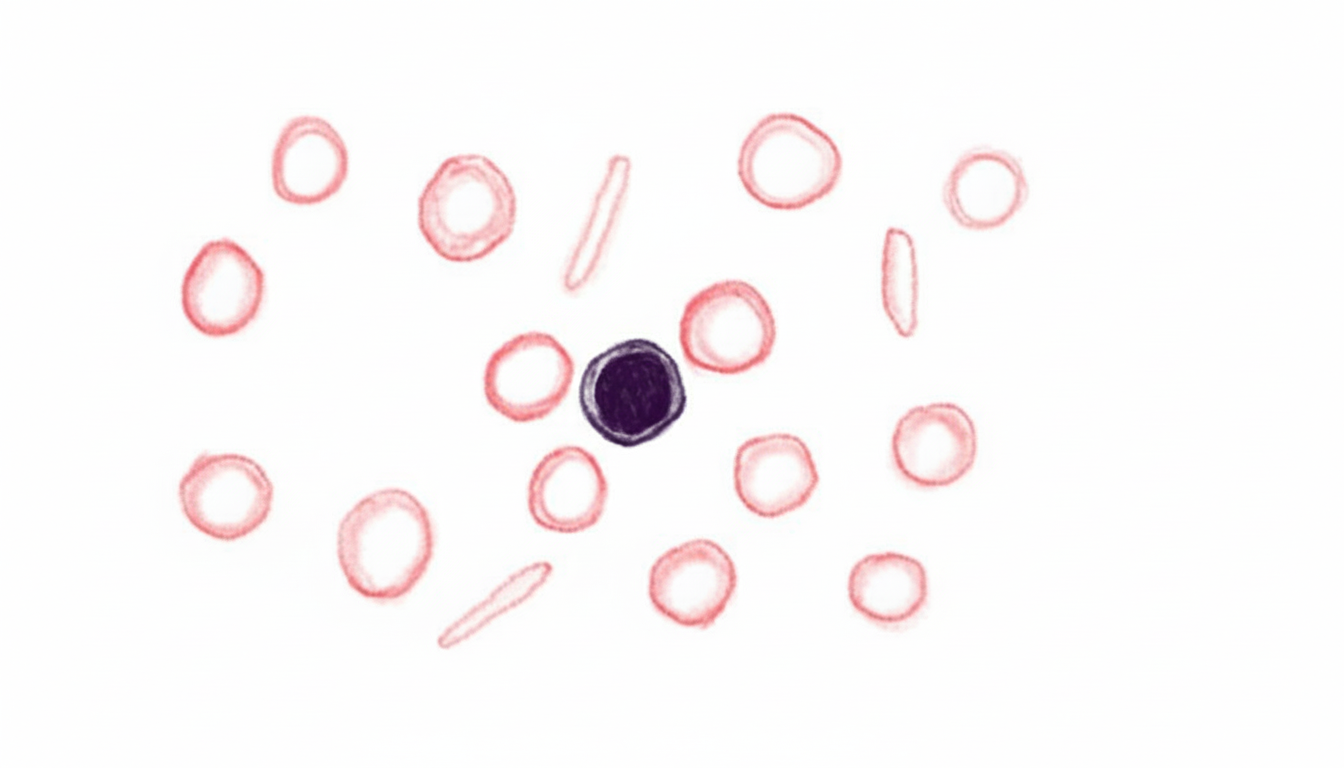
To which of the following diseases is pyruvate kinase deficiency most similar clinically?
What is the most common inherited bleeding disorder?
The Rumpel Leede test measures what?
What is the typical number of platelets in a single bag of Single Donor Platelets (SDP)?
Which of the following is a feature of disseminated intravascular coagulation (DIC)?
Practice by Chapter
Anemia Evaluation and Management
Practice Questions
Hemoglobinopathies
Practice Questions
Thalassemias
Practice Questions
Platelet Disorders
Practice Questions
Coagulation Disorders
Practice Questions
Thrombotic Disorders
Practice Questions
Leukemias
Practice Questions
Lymphomas
Practice Questions
Multiple Myeloma and Plasma Cell Disorders
Practice Questions
Myeloproliferative Neoplasms
Practice Questions
Transfusion Medicine
Practice Questions
Hematopoietic Stem Cell Transplantation
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
