Hematology — MCQs

On this page
All of the following are features of thrombotic thrombocytopenic purpura, EXCEPT:
Splenomegaly is least likely associated with which of the following conditions?
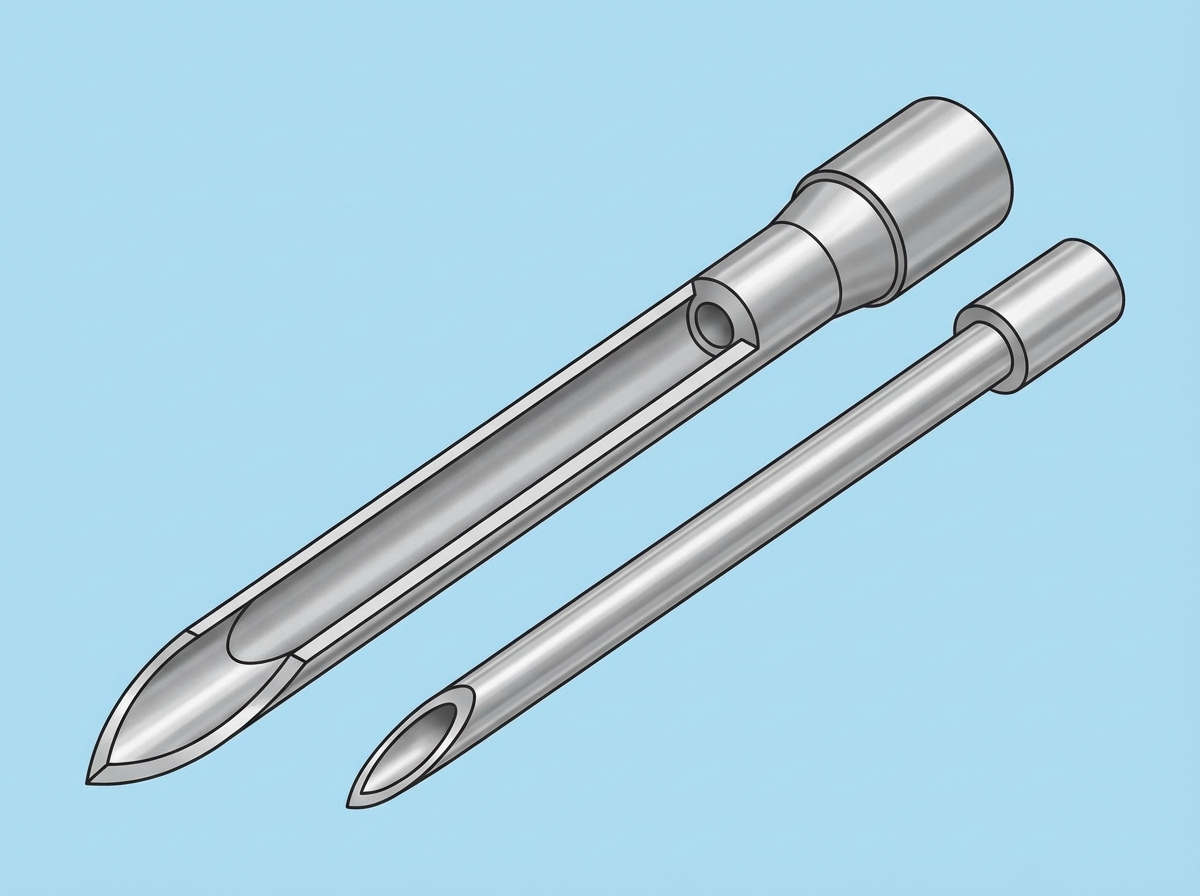
For what procedure is the instrument depicted in the diagram used?
A 20-year-old male presents with weakness, jaundice, and hematuria. He has a history of similar, self-limiting episodes and unusual substance consumption. Examination reveals mild jaundice and splenomegaly. Lab findings include increased indirect bilirubin, hemoglobinemia, hemoglobinuria, absent haptoglobin, raised LDH, hemosiderinuria, and reticulocytosis. Which peripheral blood smear finding is most likely associated with this condition?
Which condition is associated with Coomb's positive hemolytic anemia?
Which of the following will NOT show a dismal prognosis?
Elevated serum ferritin, decreased serum iron, and decreased % transferrin saturation are most consistent with which diagnosis?
A 30-year-old male, undergoing platelet apheresis for the first time, experiences tingling around the mouth and numbness in his arm. An ECG shows ST segment elevation. What is the most likely cause of these symptoms?
What is the most common subtype of adult acute lymphoblastic leukemia (ALL)?
A 58-year-old woman presents with a gastric ulcer, achlorhydria, loss of vibration sense in her lower extremities, and megaloblastic anemia. What diagnostic test would best support a diagnosis of pernicious anemia?
Practice by Chapter
Anemia Evaluation and Management
Practice Questions
Hemoglobinopathies
Practice Questions
Thalassemias
Practice Questions
Platelet Disorders
Practice Questions
Coagulation Disorders
Practice Questions
Thrombotic Disorders
Practice Questions
Leukemias
Practice Questions
Lymphomas
Practice Questions
Multiple Myeloma and Plasma Cell Disorders
Practice Questions
Myeloproliferative Neoplasms
Practice Questions
Transfusion Medicine
Practice Questions
Hematopoietic Stem Cell Transplantation
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
