Endocrinology — MCQs

On this page
Which of the following is NOT a consequence of primary hyperaldosteronism?
A 35-year-old male with a history of severe bronchial asthma for several months now complains of weight gain and the development of purple stretch marks. Physical examination showed BP 149/95 mmHg, supraclavicular fullness, thinned skin with areas of bruising, and decreased strength in proximal muscles. Laboratory investigations showed reduced serum cortisol and reduced plasma ACTH. An electrolyte workup in this patient will most likely reveal?
Which of the following is seen in secondary hyperparathyroidism?
A couple has two children and is currently unable to conceive. The father is diagnosed with hypogonadotropic hypogonadism. Which of the following statements is false regarding his condition?
A patient presents with increased serum calcium and decreased serum phosphate. What is the most likely diagnosis?
Which of the following is NOT typically seen in hypothyroidism?
What is true about thyroid storm?
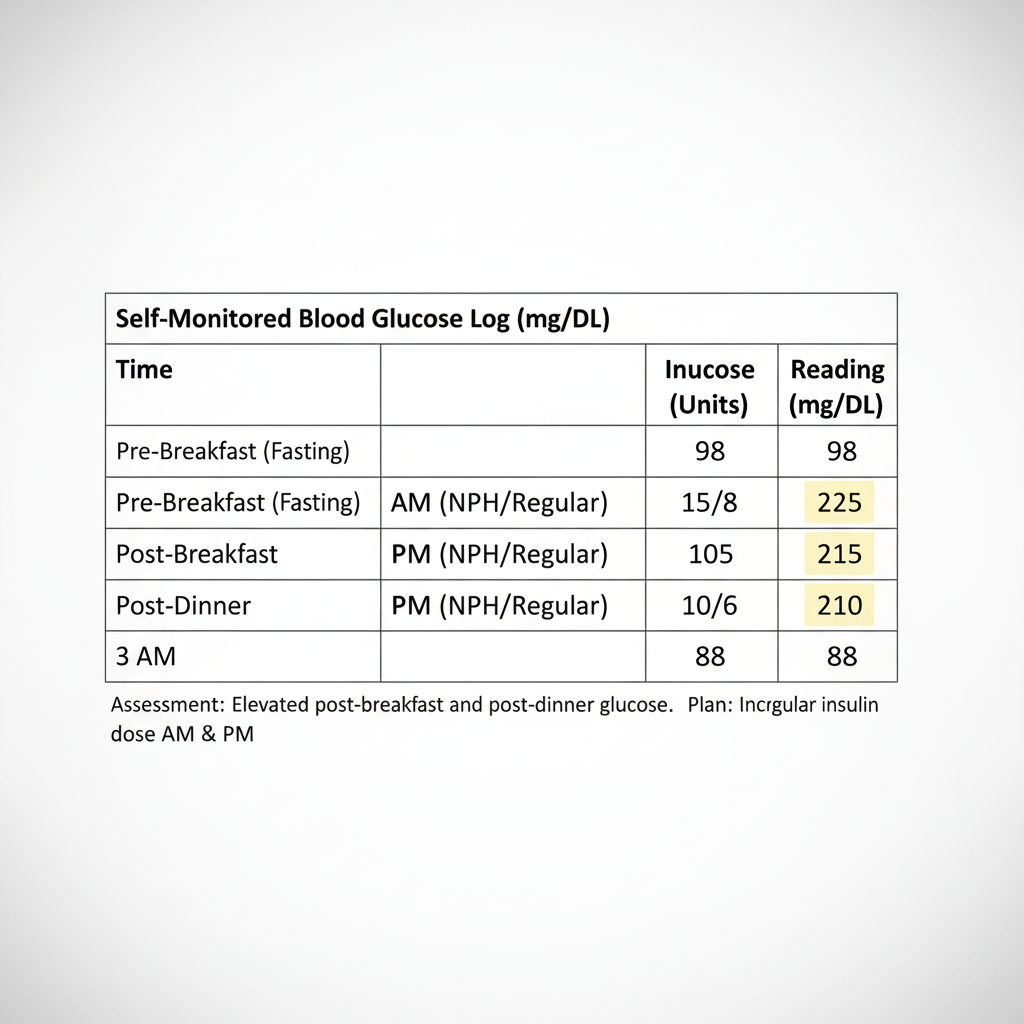
A patient with IDDM injects a mixture of NPH insulin and crystalline zinc (Regular) insulin before breakfast (at 7 AM) and before dinner (at 8 PM) each day. She reports the following average self-monitored RBS for the past week.
Which pituitary tumor is most responsive to medical therapy?
Which of the following is NOT a cause of hypercalcemia?
Practice by Chapter
Diabetes Mellitus
Practice Questions
Thyroid Disorders
Practice Questions
Adrenal Gland Disorders
Practice Questions
Pituitary Disorders
Practice Questions
Calcium and Bone Metabolism
Practice Questions
Reproductive Endocrinology
Practice Questions
Lipid Disorders
Practice Questions
Endocrine Hypertension
Practice Questions
Multiple Endocrine Neoplasia
Practice Questions
Obesity and Metabolic Syndrome
Practice Questions
Neuroendocrine Tumors
Practice Questions
Endocrine Emergencies
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
