Endocrinology — MCQs

On this page
What is the most likely underlying diagnosis?

Diabetic mellitus is characterized by:
Which of the following is true about Nelson syndrome?
Primary hyperparathyroidism is most commonly caused by?
What is the most common cause of Conn's syndrome?
All of the following can be seen in this patient except?
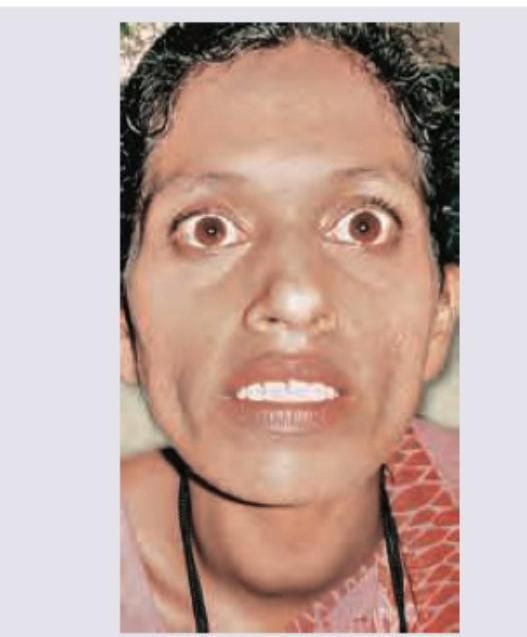
Episodic hypertension is a classical feature of which condition?
Impotency may be seen in:
Which is the first hormone secretion affected in a pituitary lesion?
A 45-year-old obese woman presents to the emergency department in a semi-comatose state. Laboratory investigations reveal K+ (5.8 mmol/L), Na+ (136 mmol/L), blood pH (7.1), HCO3 (12 mmol/L), and ketone bodies (350 mg/dl). What is the expected level of blood glucose for this patient?
Practice by Chapter
Diabetes Mellitus
Practice Questions
Thyroid Disorders
Practice Questions
Adrenal Gland Disorders
Practice Questions
Pituitary Disorders
Practice Questions
Calcium and Bone Metabolism
Practice Questions
Reproductive Endocrinology
Practice Questions
Lipid Disorders
Practice Questions
Endocrine Hypertension
Practice Questions
Multiple Endocrine Neoplasia
Practice Questions
Obesity and Metabolic Syndrome
Practice Questions
Neuroendocrine Tumors
Practice Questions
Endocrine Emergencies
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
