Endocrinology — MCQs

On this page
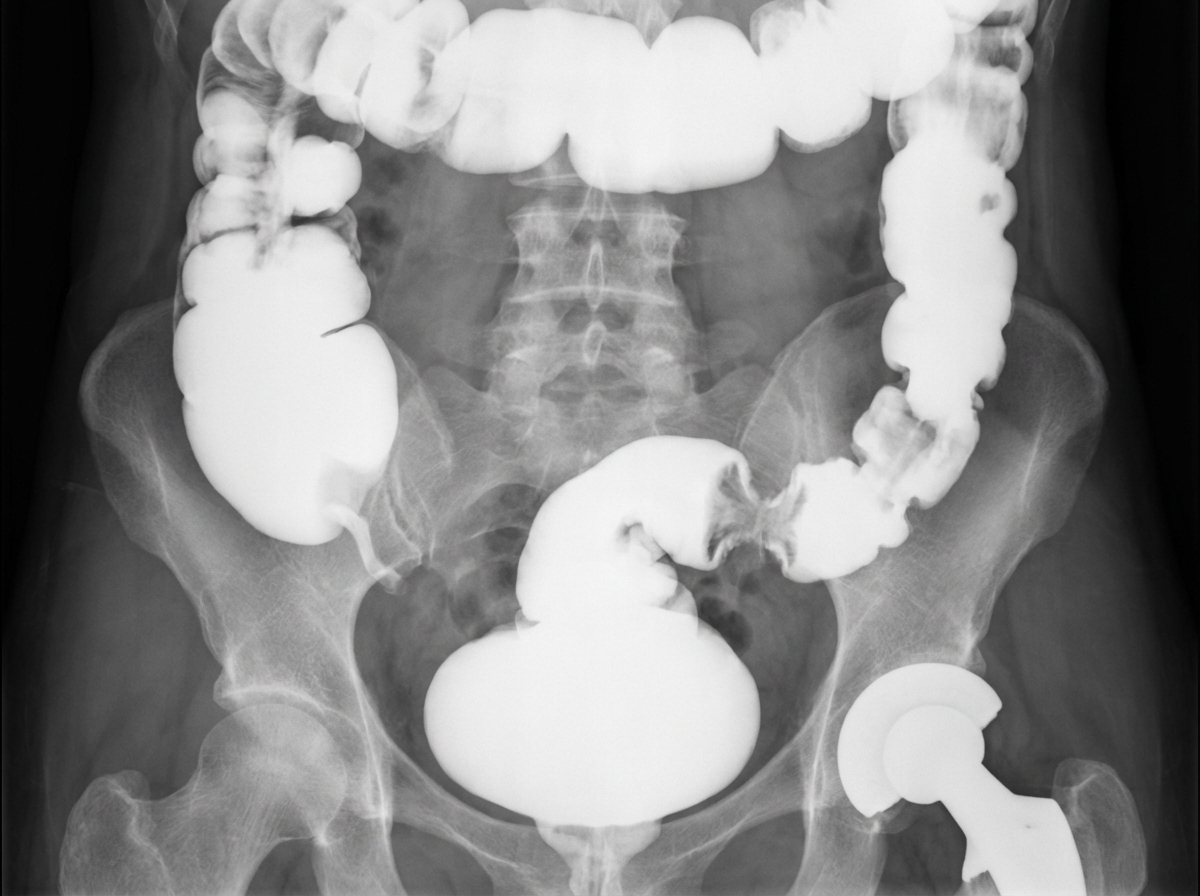
What is the most likely underlying diagnosis in this 82-year-old patient with diabetes mellitus who had undergone a total hip replacement 10 years previously?
Manifestations of endemic cretinism include:
What substance is deposited in Bronze diabetes?
Albright's syndrome includes all except?
What is the most common cause of hyperparathyroidism?
A 45-year-old patient has a Fasting Blood Sugar (FBS) of 111 mg/dL, Postprandial Blood Sugar (PPBS) of 181 mg/dL, and HbA1c of 6.1%. What is the diagnosis?
What is the characteristic feature of primary aldosteronism?
Which of the following is NOT a steroid?
Which of the following is not involved in MEN II syndrome?
Pituitary adenomas are regarded as macroadenomas when their size is:
Practice by Chapter
Diabetes Mellitus
Practice Questions
Thyroid Disorders
Practice Questions
Adrenal Gland Disorders
Practice Questions
Pituitary Disorders
Practice Questions
Calcium and Bone Metabolism
Practice Questions
Reproductive Endocrinology
Practice Questions
Lipid Disorders
Practice Questions
Endocrine Hypertension
Practice Questions
Multiple Endocrine Neoplasia
Practice Questions
Obesity and Metabolic Syndrome
Practice Questions
Neuroendocrine Tumors
Practice Questions
Endocrine Emergencies
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
