Endocrinology — MCQs

On this page
How is hyperthyroidism diagnosed?
The insulin preparation of choice in diabetic ketoacidosis is
In Sipple syndrome (MEN 2A), which of the following is typically absent?
The goals of therapy for type 1 or type 2 diabetes mellitus are all except?
Which of the following is NOT a cause of fasting hypoglycemia?
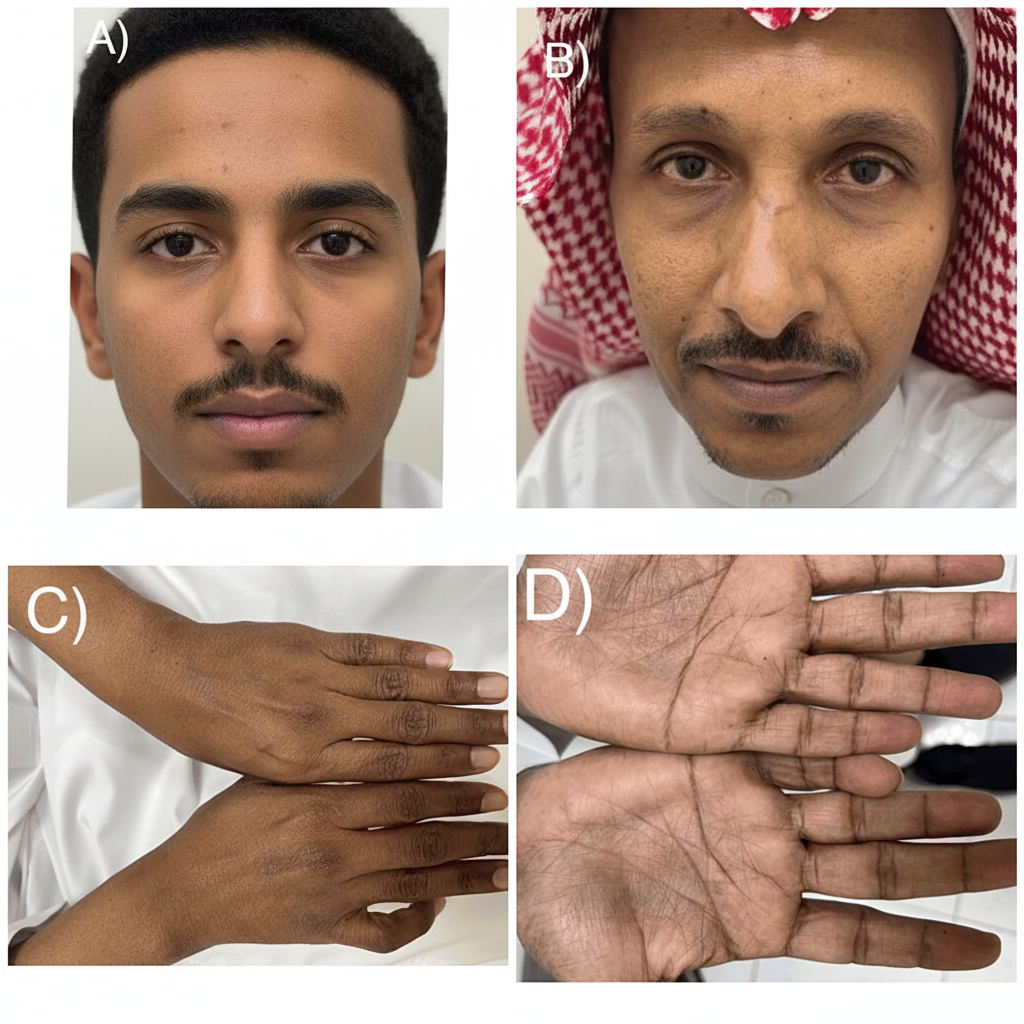
What is the most likely diagnosis in a patient with generalized weakness and the given cutaneous finding?
Liddle syndrome is characterized by
A patient presents with symptoms of hypoglycemia. Investigations reveal decreased blood glucose and increased insulin levels. A C-peptide assay shows normal levels of C-peptide. What is the most likely diagnosis?
Hypocalcemia is seen with which of the following conditions?
De Quervain's disease is:
Practice by Chapter
Diabetes Mellitus
Practice Questions
Thyroid Disorders
Practice Questions
Adrenal Gland Disorders
Practice Questions
Pituitary Disorders
Practice Questions
Calcium and Bone Metabolism
Practice Questions
Reproductive Endocrinology
Practice Questions
Lipid Disorders
Practice Questions
Endocrine Hypertension
Practice Questions
Multiple Endocrine Neoplasia
Practice Questions
Obesity and Metabolic Syndrome
Practice Questions
Neuroendocrine Tumors
Practice Questions
Endocrine Emergencies
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
