Endocrinology — MCQs

On this page
Which of the following is NOT a typical presentation of hyperthyroidism?
A 75-year-old woman presents with mild congestive heart failure 6 weeks after a myocardial infarction. She has a past history of neck surgery for parathyroid adenoma 5 years ago. Her EKG shows slow atrial fibrillation. Serum calcium is 13.0 mg/dL and urinary calcium is 300 mg/24 hr. On examination, there is a small mass in the paratracheal position behind the right clavicle. What is the appropriate management at this time?
Vitamin D-resistant rickets is associated with all of the following, except?
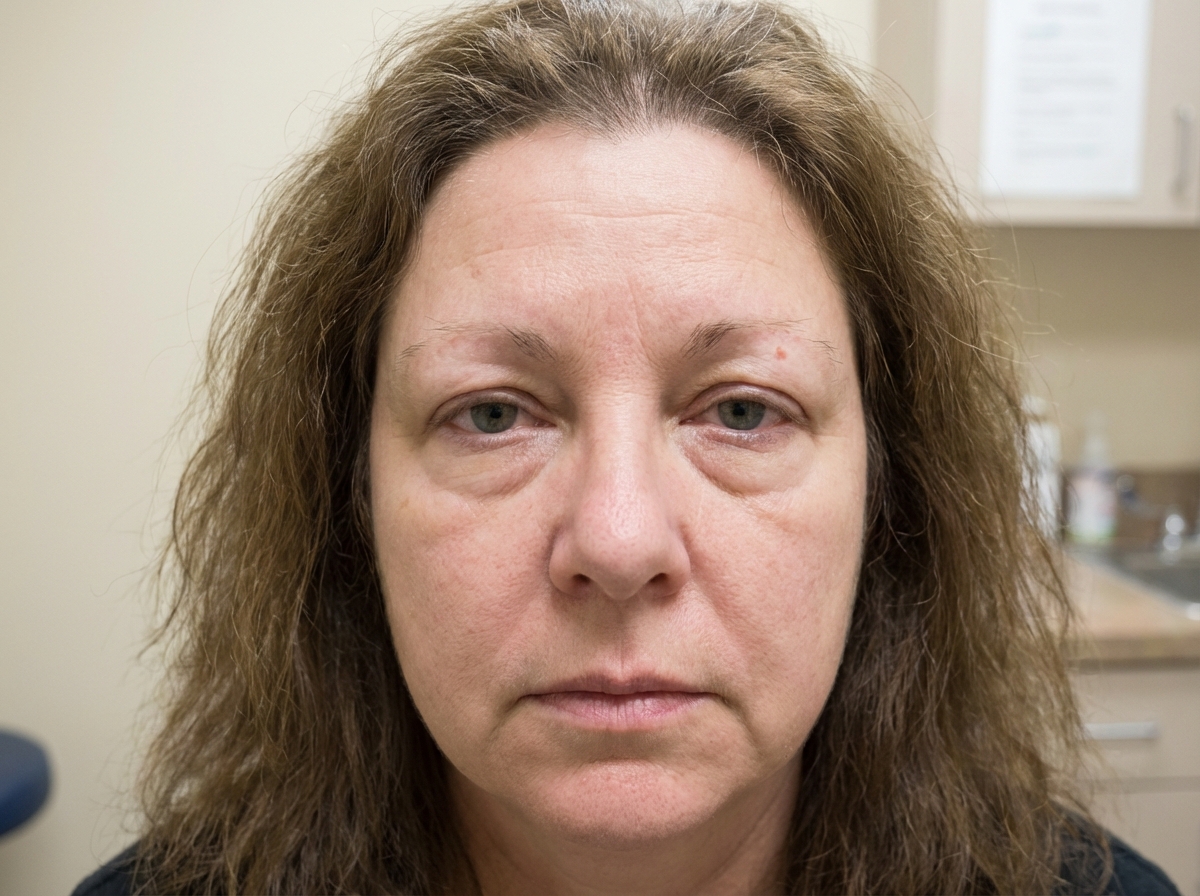
Which of the following conditions is most likely to be associated with the clinical findings shown in the image?
Galactorrhea may be associated with the use of all the following drugs except?
In hyperparathyroidism, which of the following is not typically seen?
Hypophosphatemia is seen in which of the following conditions?
What fasting blood glucose level is indicative of diabetes mellitus?
What is the immediate treatment of hypercalcemia of malignancy?
All of the following occur in adrenal deficiency except?
Practice by Chapter
Diabetes Mellitus
Practice Questions
Thyroid Disorders
Practice Questions
Adrenal Gland Disorders
Practice Questions
Pituitary Disorders
Practice Questions
Calcium and Bone Metabolism
Practice Questions
Reproductive Endocrinology
Practice Questions
Lipid Disorders
Practice Questions
Endocrine Hypertension
Practice Questions
Multiple Endocrine Neoplasia
Practice Questions
Obesity and Metabolic Syndrome
Practice Questions
Neuroendocrine Tumors
Practice Questions
Endocrine Emergencies
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
