Biostatistics — MCQs

On this page
A clinical trial is stopped early after interim analysis shows the new treatment is superior to standard care (p = 0.001). However, only 60% of planned enrollment was completed. Analyze the implications of early trial termination.
A meta-analysis of 20 studies shows heterogeneity (I² = 85%) in results examining the effectiveness of a new treatment. The overall effect size is statistically significant (p < 0.01). Analyze the implications of this heterogeneity for clinical practice.
A randomized controlled trial of a new antidepressant shows statistically significant improvement (p = 0.02) compared to placebo. However, the effect size is small (Cohen's d = 0.2), and the confidence interval barely excludes the null. Analyze the clinical implications of these findings.
A research study on a new diabetes medication reports the following: Control group event rate = 20%, Treatment group event rate = 15%, Relative risk = 0.75, Number needed to treat = 20. Analyze these results and determine what they indicate about the treatment's clinical significance.
A clinical trial comparing two medications reports a relative risk of 0.75 (95% CI: 0.60-0.90) for the primary outcome. How should this result be interpreted?
A researcher conducts a study comparing two treatments and finds a p-value of 0.08. The study had 80% power to detect a clinically meaningful difference. How should this result be interpreted?
A diagnostic test has a likelihood ratio positive (LR+) of 10 and likelihood ratio negative (LR-) of 0.1. A patient has a pre-test probability of 20% for the disease. If the test is positive, what is the approximate post-test probability?
A research study found a statistically significant result with p = 0.03. The confidence interval for the effect size is 0.5 to 2.1. How should this result be interpreted?
A new screening test for disease X has a sensitivity of 90% and specificity of 80%. In a population where the prevalence of disease X is 5%, what is the positive predictive value of this test?
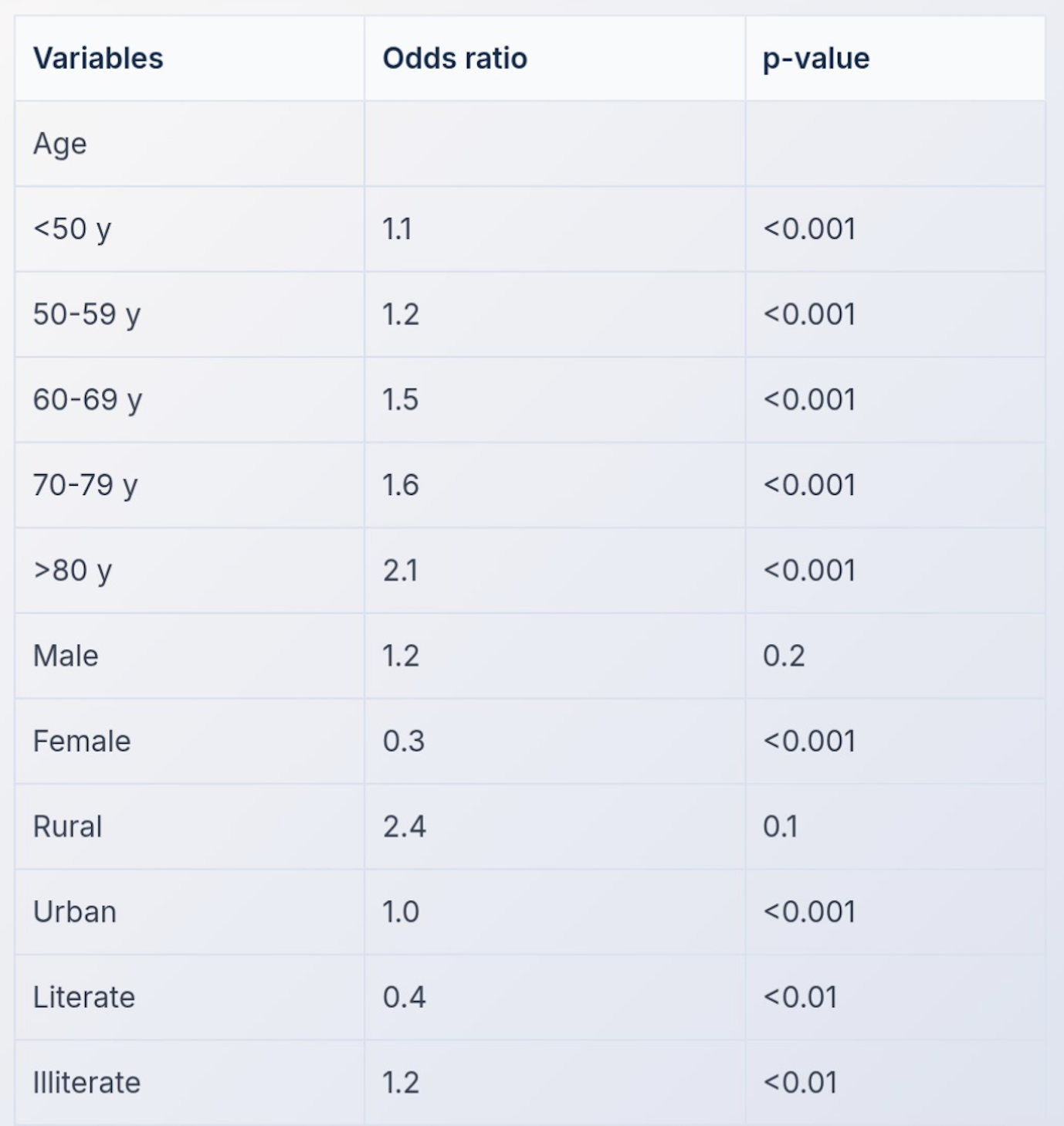
A multivariate analysis was conducted to examine the relationship between risk of developing blindness and age. The results are shown in the table below. Which of the following is true?
Practice by Chapter
Collection and Presentation of Data
Practice Questions
Measures of Central Tendency
Practice Questions
Measures of Dispersion
Practice Questions
Normal Distribution
Practice Questions
Sampling Methods
Practice Questions
Sample Size Calculation
Practice Questions
Hypothesis Testing
Practice Questions
Tests of Significance
Practice Questions
Correlation and Regression
Practice Questions
Survival Analysis
Practice Questions
Multivariate Analysis
Practice Questions
Statistical Software in Research
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
