Biostatistics — MCQs

On this page
In the estimation of statistical probability, Z score is applicable to:
The two important values necessary for describing the variation in a series of observations are:
Which one of the following tests should be applied to compare mean haemoglobin level of two groups of antenatal mothers?
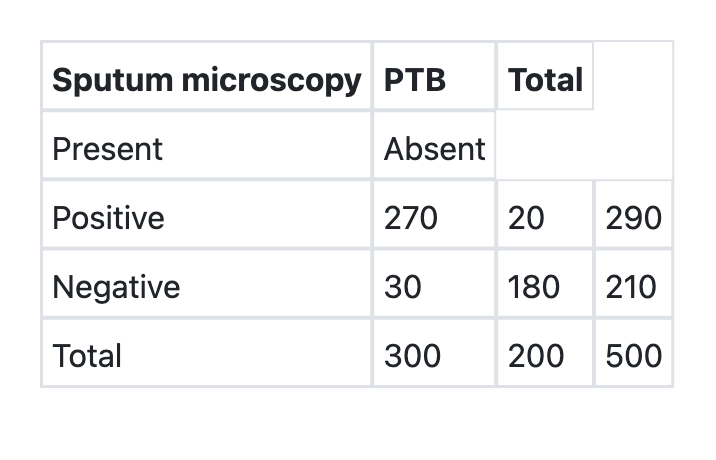
What is the specificity of sputum microscopy in detection of Pulmonary Tuberculosis (PTB) as per the information given below?
A research study comparing two groups shows a statistically significant difference (p=0.04) but the confidence interval is very wide (0.5 to 4.2). How should this result be interpreted?
A randomized controlled trial of a new diabetes medication is stopped early when interim analysis shows significant benefit. The trial enrolled only 60% of the planned participants. What is the most important consideration about this early termination?
A meta-analysis of 10 studies shows significant benefit from a new treatment (p=0.001), but there is high heterogeneity between studies (I² = 78%). Individual studies show conflicting results. How should this evidence be interpreted?
A clinical trial shows that a new cardiac medication reduces mortality from 20% to 15%. The relative risk is 0.75 and the number needed to treat is 20. How should the clinical significance be interpreted?
A researcher wants to study a new antidepressant in patients with severe depression. Standard antidepressants are available and effective. The pharmaceutical company insists on a placebo-controlled design, arguing it provides the clearest evidence of efficacy. What is the most appropriate study design?
A surgeon notices that their complication rates are higher than expected. Hospital administration suggests this might be due to taking more complex cases. Analyze the statistical concept that explains this phenomenon.
Practice by Chapter
Collection and Presentation of Data
Practice Questions
Measures of Central Tendency
Practice Questions
Measures of Dispersion
Practice Questions
Normal Distribution
Practice Questions
Sampling Methods
Practice Questions
Sample Size Calculation
Practice Questions
Hypothesis Testing
Practice Questions
Tests of Significance
Practice Questions
Correlation and Regression
Practice Questions
Survival Analysis
Practice Questions
Multivariate Analysis
Practice Questions
Statistical Software in Research
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
