Hemoglobin and Iron Metabolism — MCQs

On this page
Which of the following is an allosteric protein?
Raised serum ferritin is seen in?
Which of the following are NOT vitamin K dependent clotting factors?
Low serum haptoglobin in hemolysis is masked by which of the following conditions?
The conversion of Fe+2 to Fe+3 is called which reaction?
Which of the following statements is true regarding the binding of O2 to hemoglobin?
What is true of iron?
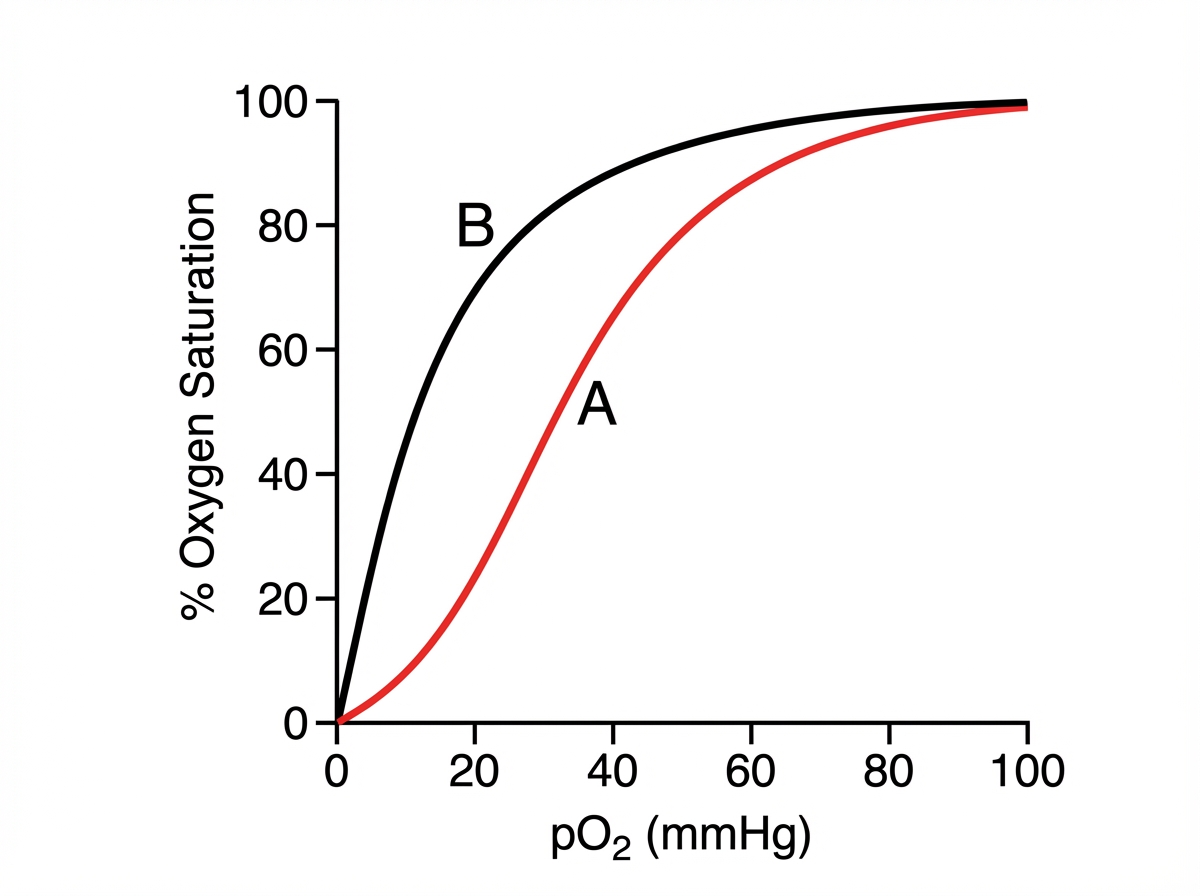
Study the given oxygen-hemoglobin dissociation curve and comment on the finding shown, identifying which protein corresponds to curve A and which to curve B.
Hemoglobin electrophoresis in SC A (sickle cell anemia) shows the presence of which hemoglobin?
Ceruloplasmin contains which of the following metals?
Practice by Chapter
Hemoglobin Structure and Function
Practice Questions
Oxygen Transport and Oxygen-Hemoglobin Dissociation Curve
Practice Questions
Hemoglobin Variants and Hemoglobinopathies
Practice Questions
Thalassemias
Practice Questions
Methemoglobin and Abnormal Hemoglobins
Practice Questions
Hemoglobin Synthesis
Practice Questions
Heme Synthesis and Porphyrias
Practice Questions
Iron Absorption and Transport
Practice Questions
Iron Storage and Recycling
Practice Questions
Disorders of Iron Metabolism
Practice Questions
Anemia: Biochemical Aspects
Practice Questions
Biochemistry of Hemostasis
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
