Hemoglobin and Iron Metabolism — MCQs

On this page
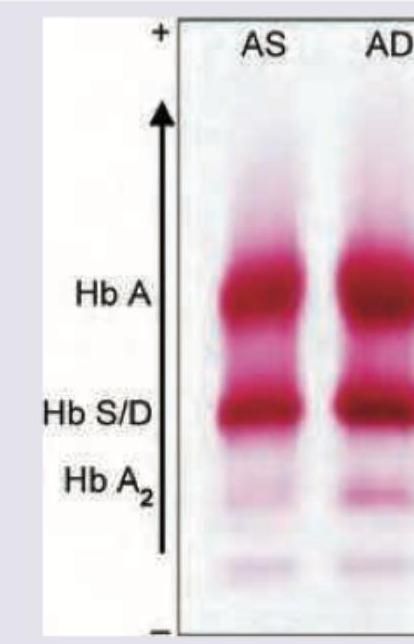
The shown pattern in electrophoresis is due to:
The single most sensitive tool for evaluating the iron status in an individual is
Which of the following interfere with iron absorption?
Reagent used in Apt test
A useful screening test for lead is measurement of which of the following?
All are involved in bilirubin metabolism except?
Hepcidin inhibits which of the following?
Which TCA intermediate is used in haem synthesis?
Which factor controls the release of iron from macrophages?
Which is not a product of heme catabolism?
Practice by Chapter
Hemoglobin Structure and Function
Practice Questions
Oxygen Transport and Oxygen-Hemoglobin Dissociation Curve
Practice Questions
Hemoglobin Variants and Hemoglobinopathies
Practice Questions
Thalassemias
Practice Questions
Methemoglobin and Abnormal Hemoglobins
Practice Questions
Hemoglobin Synthesis
Practice Questions
Heme Synthesis and Porphyrias
Practice Questions
Iron Absorption and Transport
Practice Questions
Iron Storage and Recycling
Practice Questions
Disorders of Iron Metabolism
Practice Questions
Anemia: Biochemical Aspects
Practice Questions
Biochemistry of Hemostasis
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
