Genetic Disorders and Biochemical Pathology — MCQs

On this page
Which of the following is NOT a disorder due to peroxisomal abnormalities?
Lack of a specific lysosomal hydrolase for glycoproteins will most likely cause which of the following conditions?
All the following are features of Von Gierke disease except?
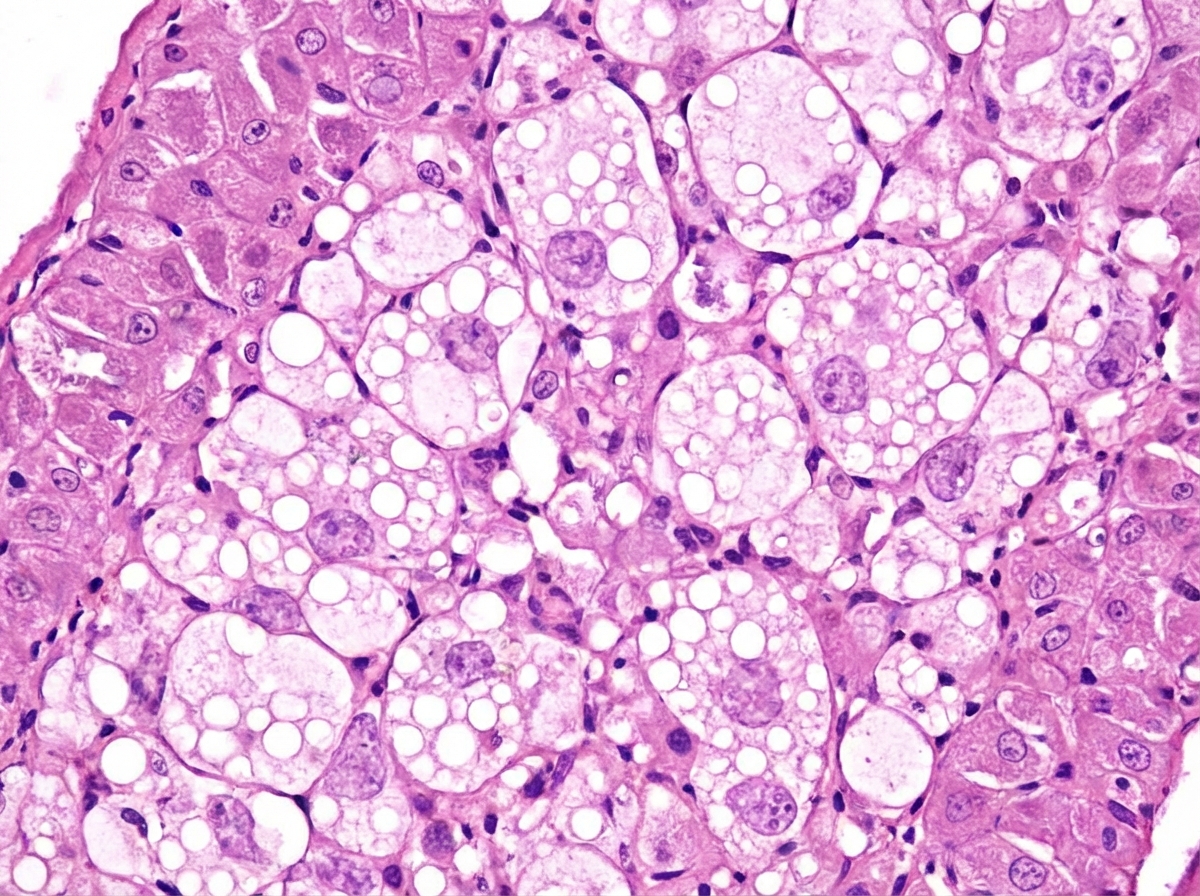
Which disease is characterized by the following histopathological finding?
Which of the following conditions causes hyperuricemia due to both increased production and decreased excretion?
A 1-year-old child presents with mental retardation, blond hair, and convulsions. What is the most probable diagnosis?
Porphyrins absorb light in which wavelength range?
A 4-year-old girl presents with sudden onset of right hip pain. Examination reveals a dislocated right hip. She can bend her thumb backward to touch her forearm, and her skin is extraordinarily stretchable. Spinal radiographs show marked lateral and anterior curvature. She develops retinal detachments later in childhood. A sibling is similarly affected, and a mutation in tenascin-X is identified. What is the most likely underlying cause for this child's findings?
Adrenoleukodystrophy is characterized by which mode of inheritance?
Menkes Kinky hair syndrome is characterized by congenital deficiency of which of the following?
Practice by Chapter
Single Gene Disorders
Practice Questions
Biochemical Diagnosis of Genetic Disorders
Practice Questions
Inborn Errors of Metabolism
Practice Questions
Lysosomal Storage Diseases
Practice Questions
Glycogen Storage Diseases
Practice Questions
Disorders of Lipoprotein Metabolism
Practice Questions
Disorders of Purine and Pyrimidine Metabolism
Practice Questions
Hemoglobinopathies
Practice Questions
Porphyrias
Practice Questions
Biochemical Markers for Disease Diagnosis
Practice Questions
Newborn Screening for Genetic Disorders
Practice Questions
Enzyme Replacement Therapy
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
