Genetic Disorders and Biochemical Pathology — MCQs

On this page
A 17-year-old with type 1 diabetes presents with diabetic ketoacidosis after a weekend camping trip. He is noted to be jaundiced, and the plasma has a red color. Which of the following is a product of the enzyme for which this patient has a deficiency?
A young male presented with an X-linked recessive disorder characterized by hyperuricemia and mild mental retardation. What is the underlying biochemical defect?
Which of the following inborn errors of metabolism is associated with mental retardation?
The following pedigree is associated with which of the following conditions?
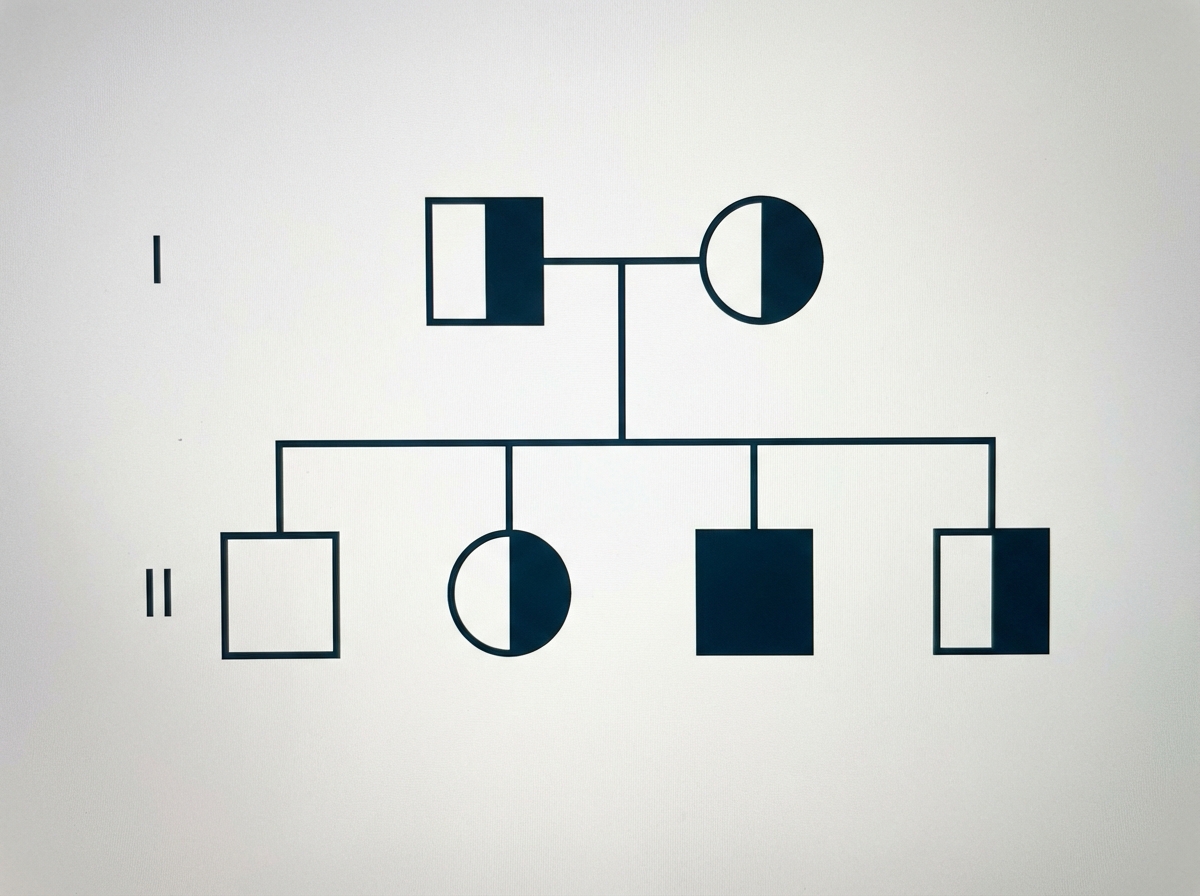
Alkaptonuria is a metabolic disease caused by a defect in or lack of homogentisic acid oxidase, which leads to what?
All of the following are examples of predominantly unconjugated hyperbilirubinemia, except?
In sickle cell anemia, which amino acid replaces glutamic acid?
A young boy presents with difficulty in getting up from a sitting position and is diagnosed with Duchenne's muscular dystrophy. Which of the following statements is true regarding a mutation in the promoter region of the dystrophin gene?
NARP syndrome is a part of which of the following group of disorders?
Zinc transporter 8 (ZnT8) antibody is seen in which of the following conditions?
Practice by Chapter
Single Gene Disorders
Practice Questions
Biochemical Diagnosis of Genetic Disorders
Practice Questions
Inborn Errors of Metabolism
Practice Questions
Lysosomal Storage Diseases
Practice Questions
Glycogen Storage Diseases
Practice Questions
Disorders of Lipoprotein Metabolism
Practice Questions
Disorders of Purine and Pyrimidine Metabolism
Practice Questions
Hemoglobinopathies
Practice Questions
Porphyrias
Practice Questions
Biochemical Markers for Disease Diagnosis
Practice Questions
Newborn Screening for Genetic Disorders
Practice Questions
Enzyme Replacement Therapy
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
