Detoxification Pathways — MCQs

On this page
Which of the following is the main enzyme responsible for the activation of xenobiotics?
Which substance is involved in the conjugation process in the liver?
Which of the following acts as a major intracellular antioxidant and helps in detoxifying reactive oxygen species?
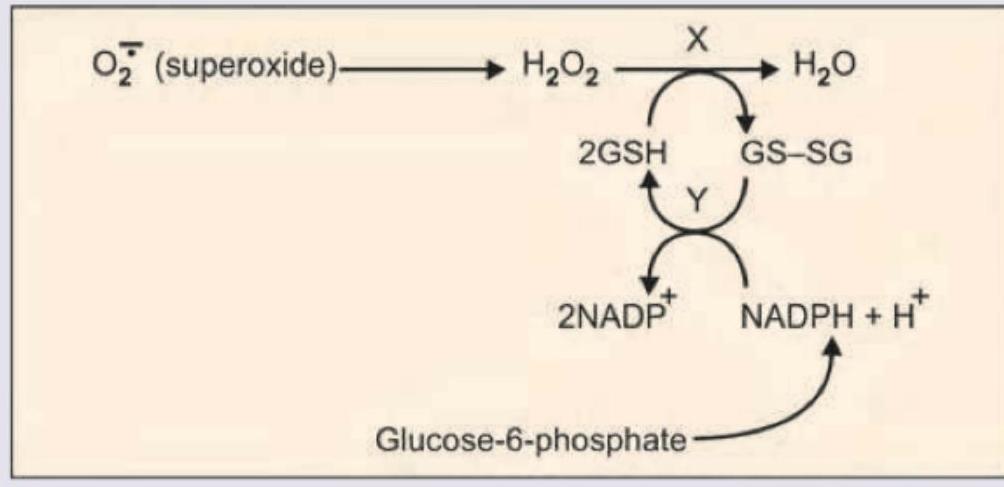
Which is correct about the image shown below?
Bilirubin conjugation with glucuronic acid has the following properties -
Glucuronide reaction is seen in
Ammonia is detoxified in brain to :
The following are major free radical scavengers except:
Bile salts undergo conjugation for enhanced solubility:
Glutathione is primarily used to detoxify which reactive oxygen species?
Practice by Chapter
Phase I Reactions: Cytochrome P450 System
Practice Questions
Phase II Conjugation Reactions
Practice Questions
Glutathione and Detoxification
Practice Questions
Drug Metabolism
Practice Questions
Metabolism of Xenobiotics
Practice Questions
Alcohol Metabolism
Practice Questions
Free Radical Generation and Antioxidant Defense
Practice Questions
Antioxidant Enzymes
Practice Questions
Detoxification of Heavy Metals
Practice Questions
Ammonia Detoxification Pathways
Practice Questions
Bilirubin Metabolism and Jaundice
Practice Questions
Biotransformation in Liver Disease
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
